
Prescribe Acthar Gel now
Start the referral process for your appropriate patients
Zand L, Canetta P, Lafayette R, Aslam N, Jan N, Sethi S, Fervenza FC—Kidney International Reports, 2020
To evaluate proteinuria reduction and remission rates in patients at high risk of progression 6 months after completing Acthar Gel treatment
*Patients with proteinuria >1 g/24 h are at high risk of progression to end-stage renal disease (ESRD).
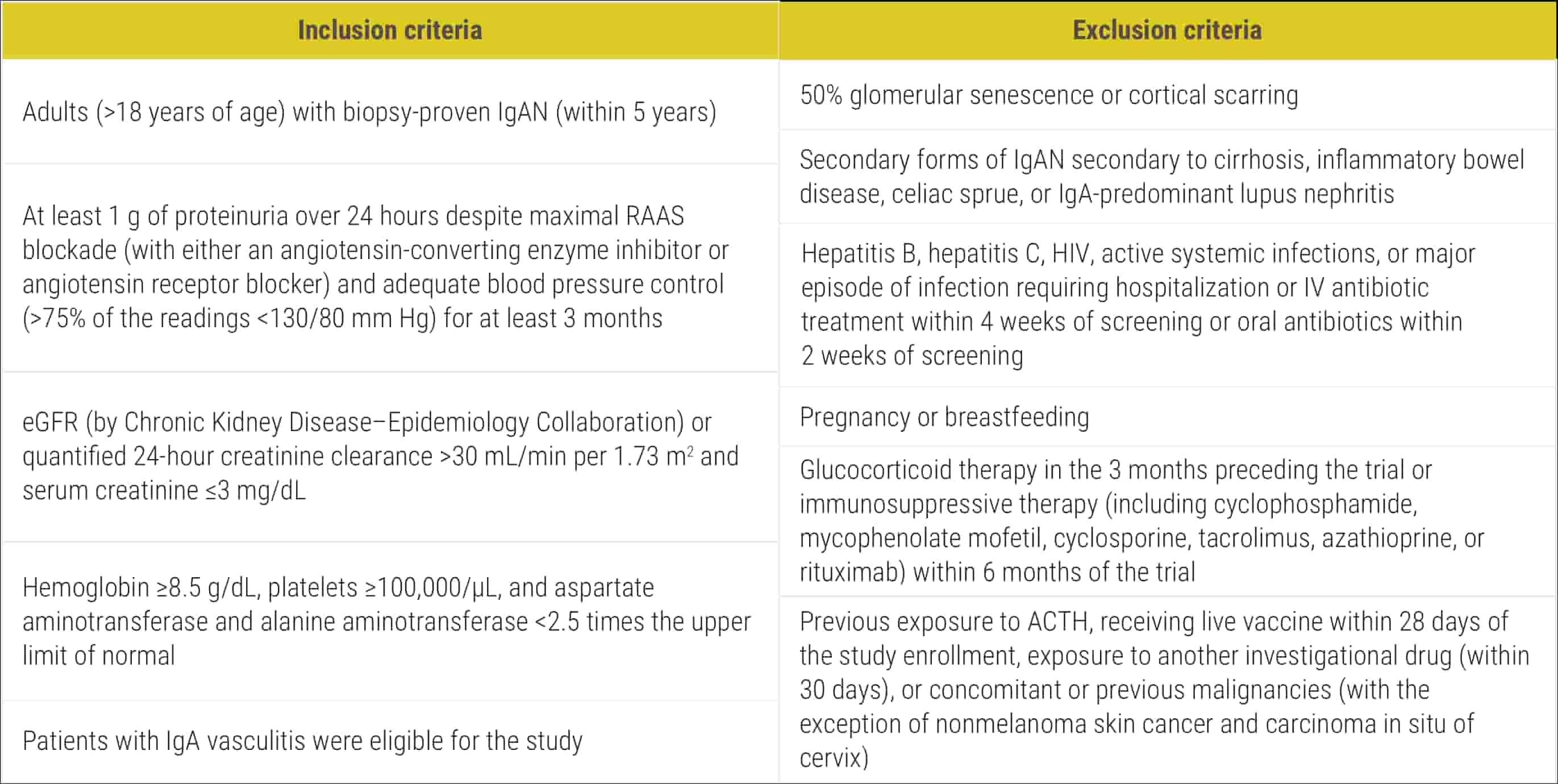
ACTH=adrenocorticotropic hormone; IV=intravenous; RAAS=renin-angiotensin-aldosterone system.
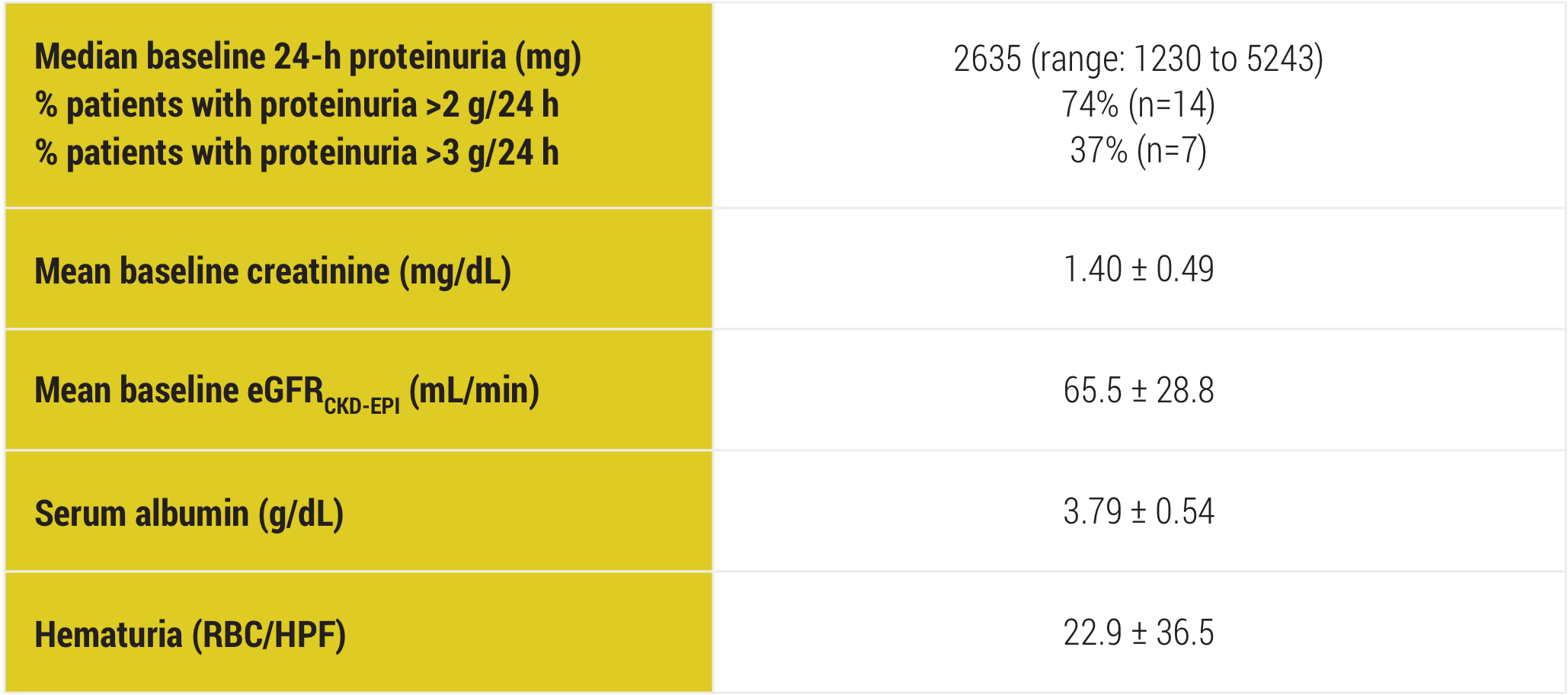
CKD-EPI=Chronic Kidney Disease-Epidemiology Collaboration; RBC/HPF=red blood cells per high-power field.
After initiation of Acthar Gel treatment, patients were seen at 1, 3, 6, 9, and 12 months for comprehensive evaluation
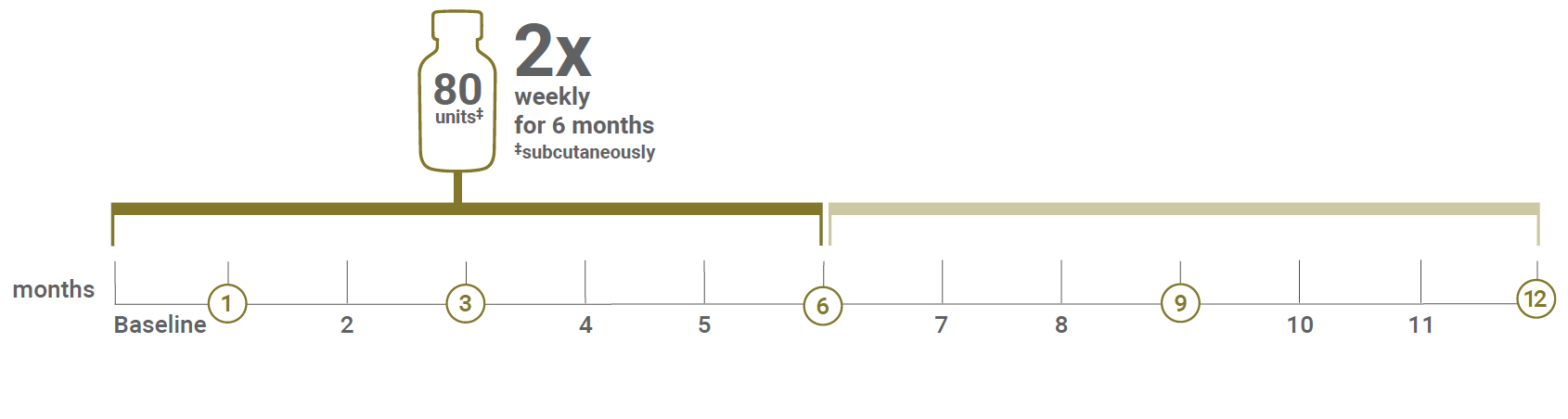
During each visit, patients completed a comprehensive physical examination, evaluation for adverse events, routine CBC, serum chemistry, HbA1c, urine analysis with microscopy, and timed urine studies for evaluation of proteinuria and creatinine clearance.
CBC=complete blood count; HbA1c=glycated hemoglobin.
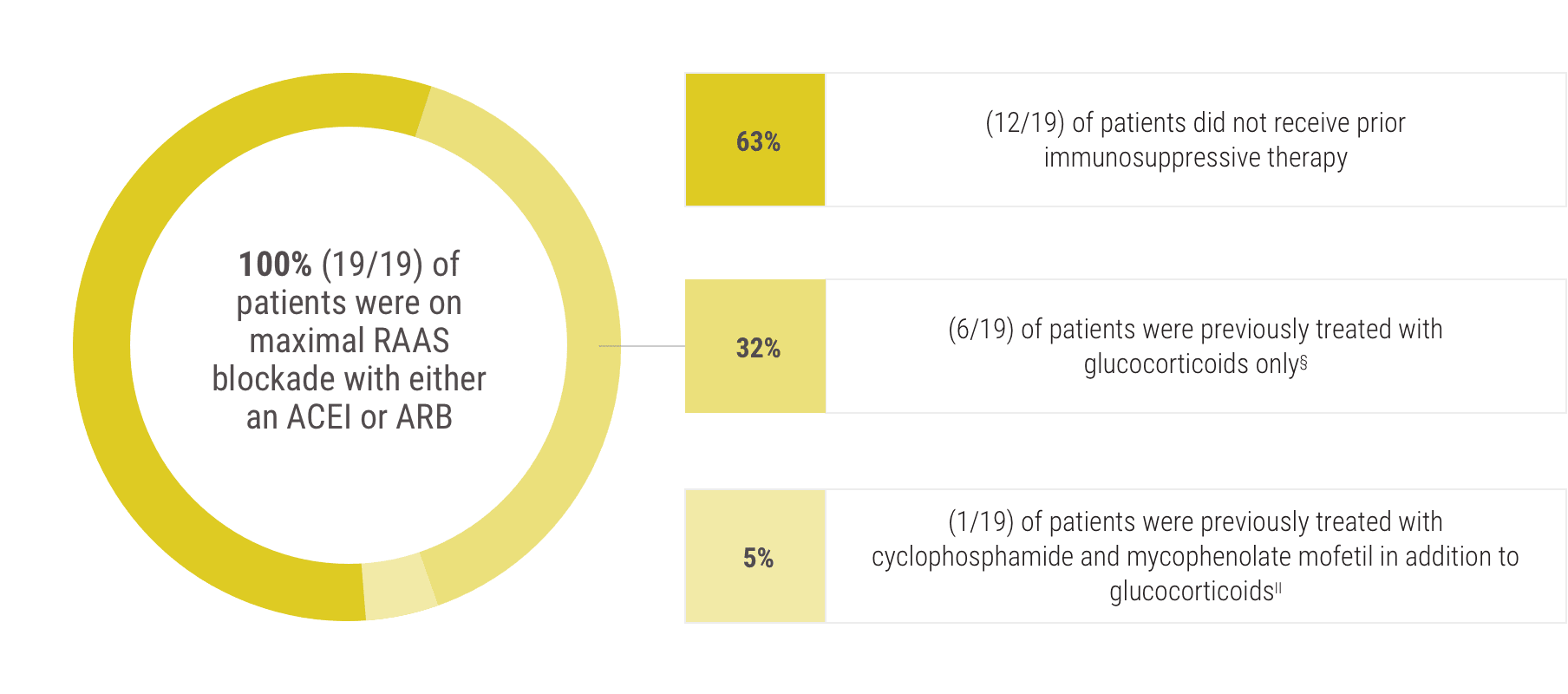
ACEI=angiotensin-converting enzyme inhibitor; ARB=angiotensin II receptor blocker.
§Treatment with glucocorticoids was stopped 3 months before enrollment in the study.
||Treatment with cyclophosphamide, mycophenolate mofetil, and glucocorticoids was stopped 6 months before enrollment in the study.
Median change from baseline in proteinuria at 6 and 12 months following Acthar Gel treatment1
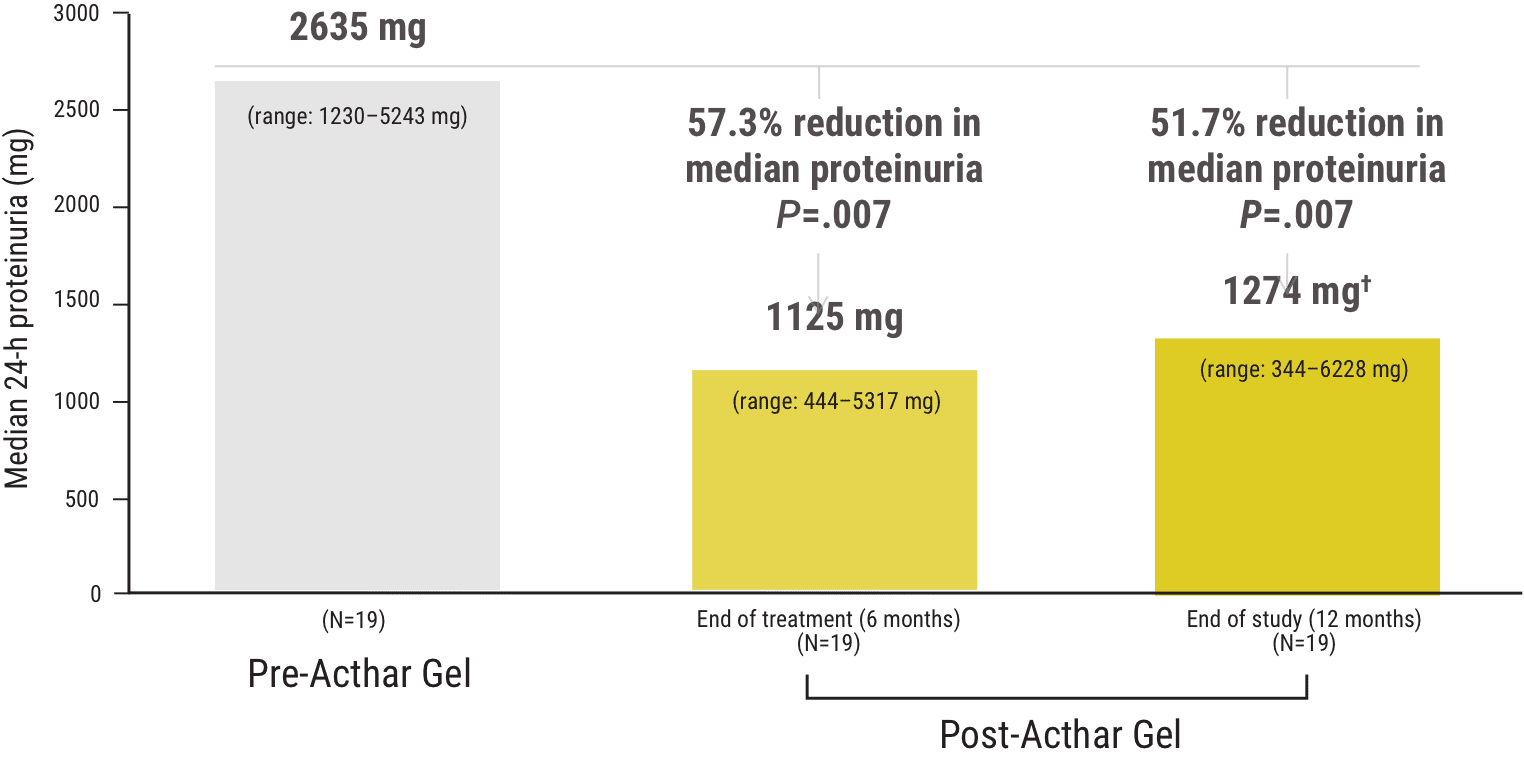
Of the 8 patients who achieved partial remission‡, median 24-hour proteinuria was recorded at 6 and 12 months following the start of Acthar Gel treatment:
†Median proteinuria at 12 months was not significantly different compared with proteinuria at 6 months (P=.9).
‡Partial remission was defined as >50% reduction in proteinuria and ≤25% decrease in eGFR from baseline.
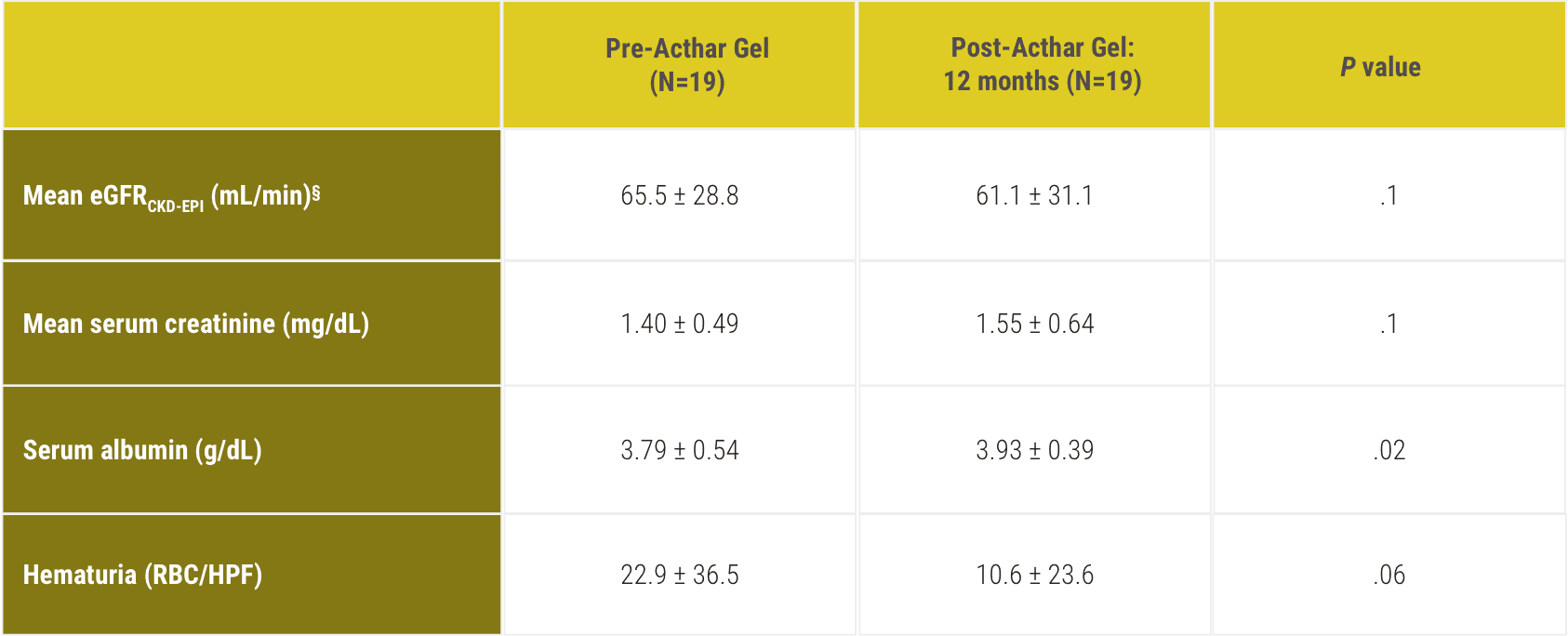
CKD-EPI=Chronic Kidney Disease-Epidemiology Collaboration; eGFR=estimated glomerular filtration rate; RBC/HPF=red blood cells per high-power field.
§eGFR was not significantly different at 12-month follow-up compared with baseline.
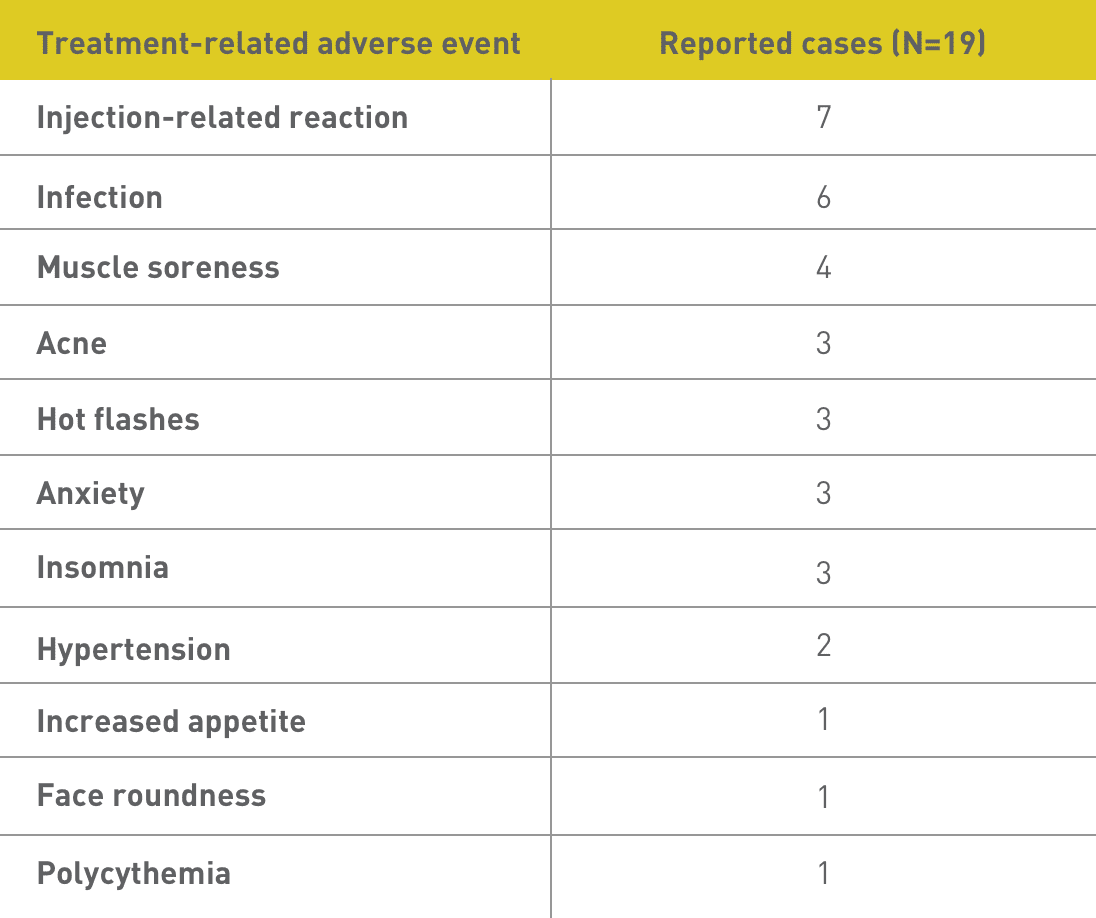
No patients in this study discontinued Acthar Gel treatment, and there were no reported cases of hyperglycemia

Start the referral process for your appropriate patients

See additional dosing information from clinical experience with Acthar Gel
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References: