
Prescribe Acthar Gel now
Start the referral process for your appropriate patients
Kaplan J, Miller T, Baker M, Due B, Zhao E—Frontiers in Neurology, 2020
Disclosure statement: Funding to support this study was provided by Mallinckrodt Pharmaceuticals.
To characterize the patient population receiving Acthar Gel for the treatment of acute MS relapse and describe their treatment patterns, MS exacerbation recovery, and safety outcomes
AE=adverse event; EDSS=Expanded Disability Status Scale; ITT=intent-to-treat; MSIS-29v1=Multiple Sclerosis Impact Scale Version 1; SAE=serious adverse event.
*Demographics, medical history, disease-modifying therapies (DMTs) (prior 2 years), and concomitant medications collected at study enrollment as well as MSIS-29v1 and EDSS.

Baseline characteristics of patients who received Acthar Gel1

Approximately two-thirds of the patients reported using concomitant DMTs at some point during the study period
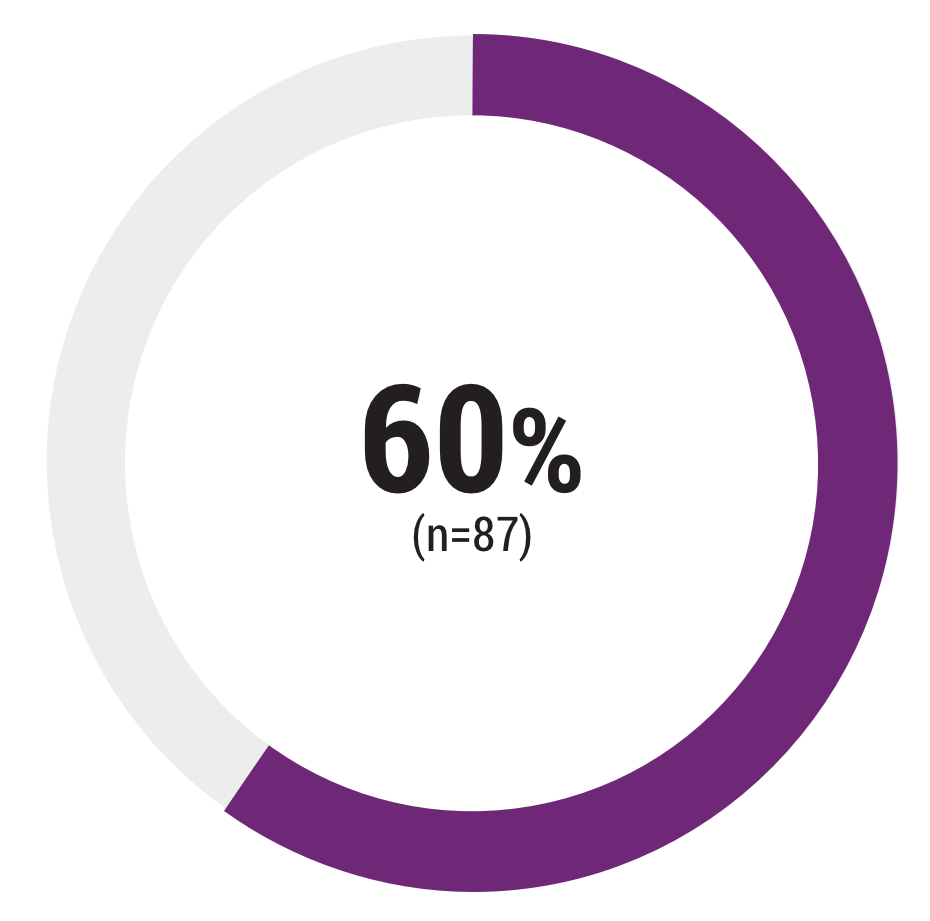
Percentage of patients that had a physician-reported history of a lack of sufficient treatment response, intolerance due to side effects, or IV access problems associated with previous treatment with high-dose glucocorticoids (eg, IVMP) for an MS relapse
IV=intravenous; IVMP=intravenous methylprednisolone.
Some patients with MS relapses experiencing recurring disease achieved clinically meaningful improvements in the MSIS-29v1 at 2 months—and sustained them at 6 months1,3,4
Mean change from baseline in the MSIS-29v1 scale1
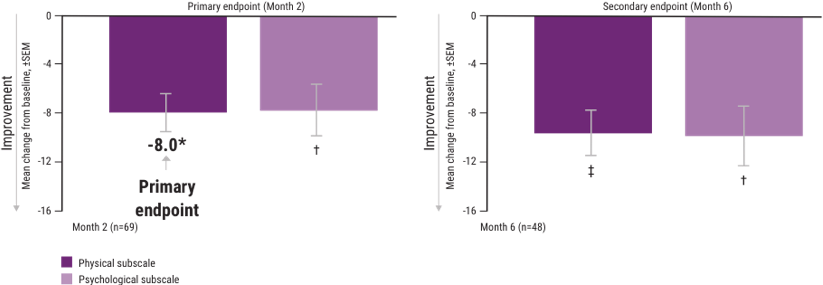
A change in score of ≥8 on the physical scale is considered clinically meaningful.5,6
ITT=intent-to-treat; MSIS-29v1=Multiple Sclerosis Impact Scale Version 1; SEM=standard error of the mean.
*P<.001.
†P<.01.
‡P<.0001.
Note: P values are based on Wilcoxon signed rank tests (ITT population) compared with baseline.
CGI-I=Clinical Global Impressions-Improvement; EDSS=Expanded Disability Status Scale.
Mean change from baseline in the EDSS scale1

Mean scores decreased at 2 and 6 months from a baseline EDSS score of 3.9.
‡P <.0001.
Note: P values are based on Wilcoxon signed rank tests (ITT population) compared with baseline.
In a post hoc analysis evaluating the number of doses, greater improvements in MSIS-29v1 and EDSS were observed in some patients taking >5 doses of Acthar Gel1
As approved by the FDA, daily intramuscular or subcutaneous doses of 80 to 120 units of Acthar Gel may be administered for 2 to 3 weeks. It may be necessary to taper the dose.9
Mean change from baseline in the MSIS-29v1 physical subscale, by number of doses administered1
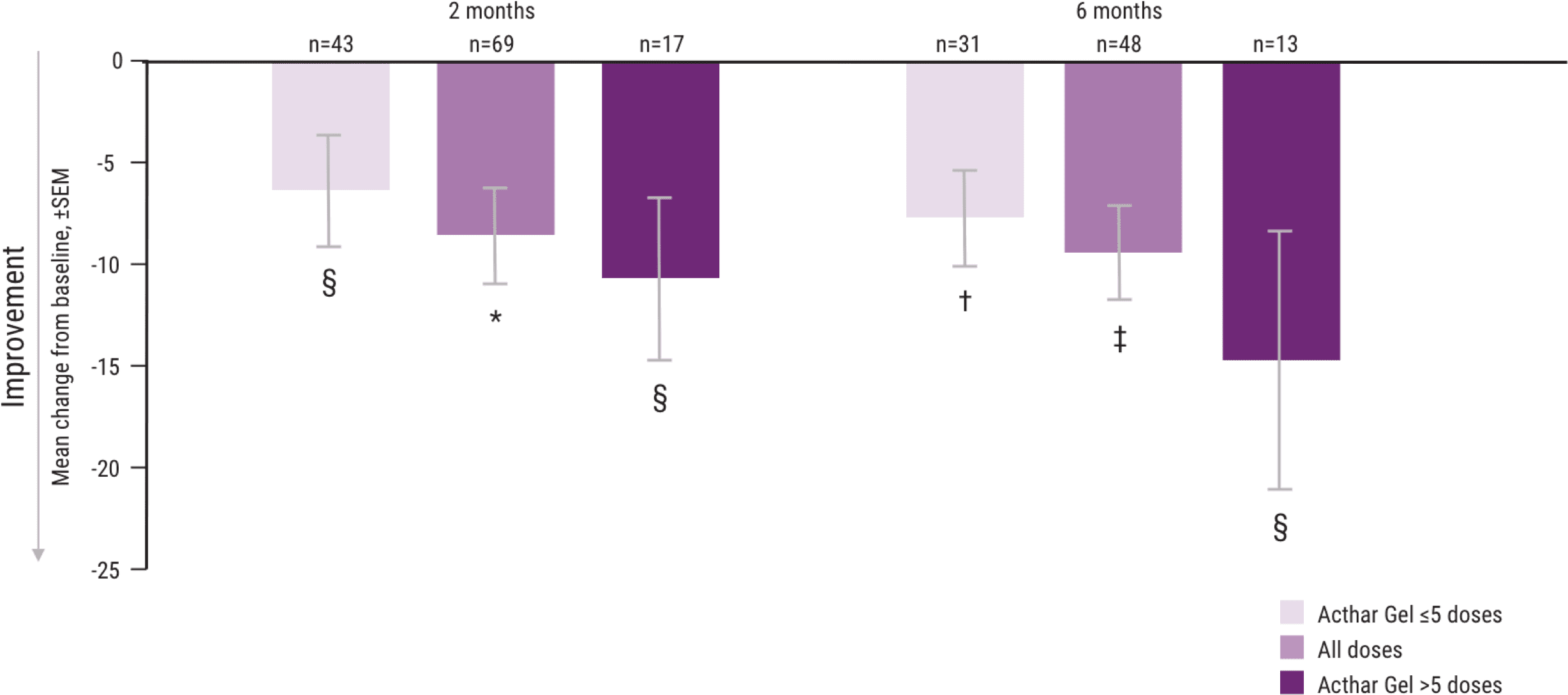
*P <.001.
†P <.01.
‡P <.0001.
§P <.05.
Note: P values are based on Wilcoxon signed rank tests (ITT population) compared with baseline.
Mean change from baseline in the EDSS scale, by number of doses administered1
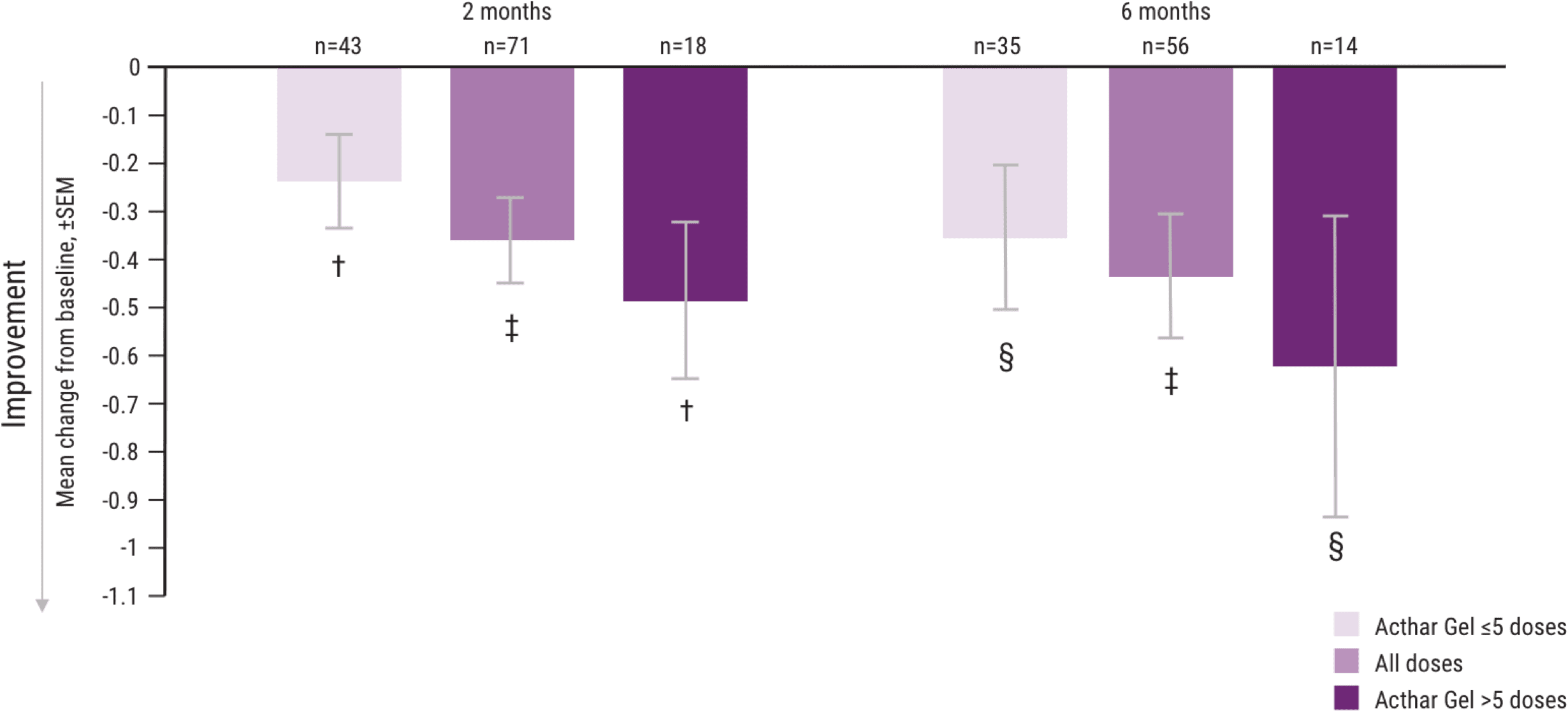
†P <.01.
‡P <.0001.
§P <.05.
Note: P values are based on Wilcoxon signed rank tests (ITT population) compared with baseline.
Acthar Gel dosing information was collected from patient self-reports.1
AEs and SAEs were consistent with the published label
AE=adverse event; SAE=serious adverse event.

Start the referral process for your appropriate patients

See additional dosing information from clinical experience with Acthar Gel
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References: