
Prescribe Acthar Gel now
Start the referral process for your appropriate patients
Anesi SD, Chang PY, Maleki A, Stephenson A, Montieth A, Filipowicz A, Syeda S, Asgari S, Walsh M, Metzinger JL, Foster S—Journal Ophthalmic and Vision Research, 2021
Disclosure statement: Funding to support this study was provided by Mallinckrodt Pharmaceuticals. Some authors of this study have financial and/or employment relationships with Mallinckrodt Pharmaceuticals.
To evaluate Acthar Gel as a treatment for patients with noninfectious retinal vasculitis who were treatment-naïve or had failed to respond to other therapies

RVSS is an objective measurement of 5 regions, assessing the severity of leakage using a predefined scale. The 5 regions assessed included:
RVSS score breakdown
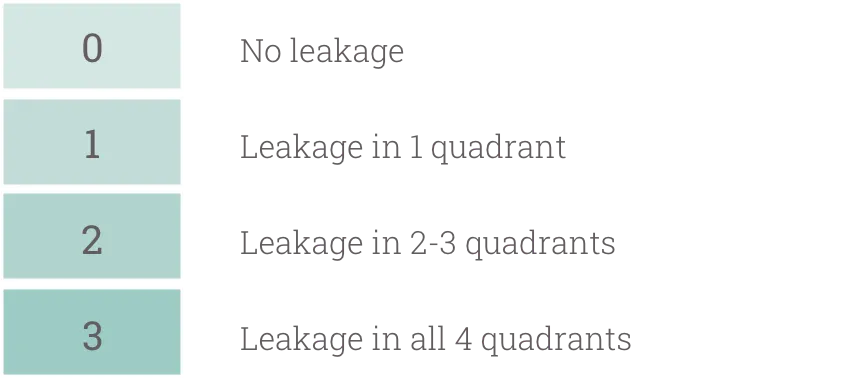


Response level is a subjective measurement determined by investigators.*
Response level breakdown

*Scores were determined by investigators independently without referencing other clinical data collected at the visit. Scores were compared between investigators and any discrepancies were discussed until an acceptable score was agreed upon.
†The same categories were used for the negative scores.
Key inclusion and exclusion criteria


Vasculature involvement, n (%)
Retinal vasculitis, n (%)
Patients in the efficacy analysis were not treated with any systemic corticosteroids during the study period.
Results were seen at Week 12 and continued through Week 24
§Findings included anterior chamber cell and flare as well as vitreous cell and haze.
IITo assess for potential toxicity to the medication.
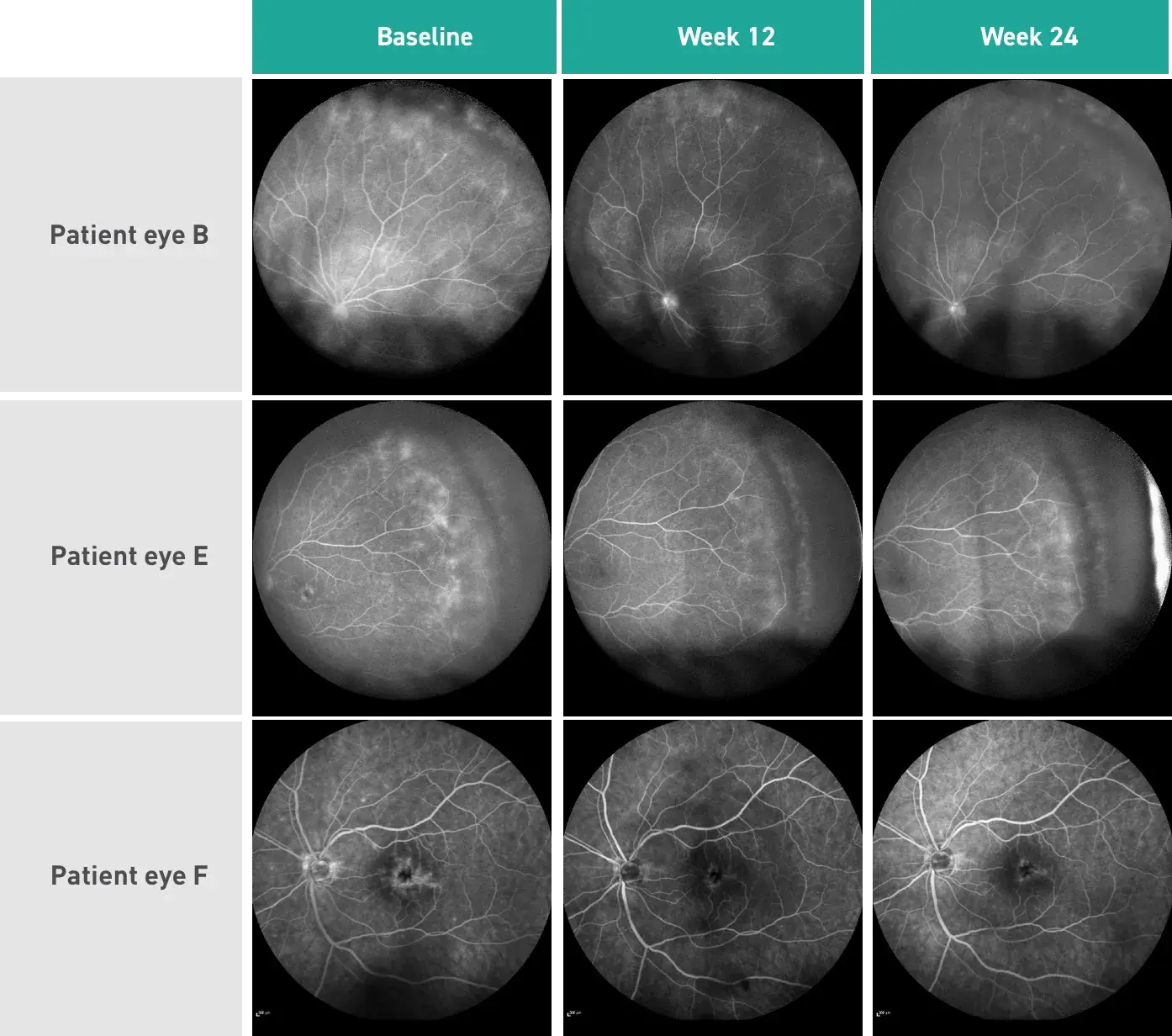
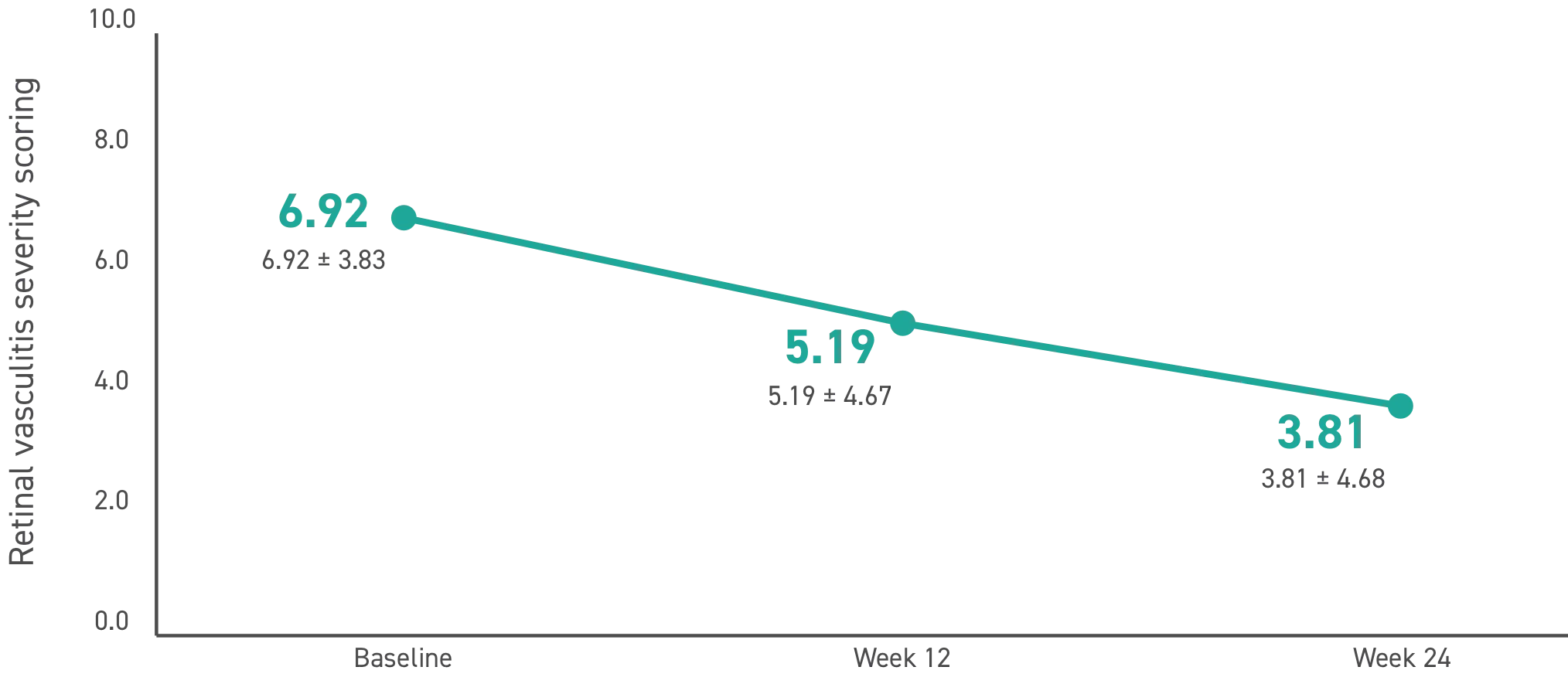
P<0.001 for the 24-week change of study indices by repeated measures analysis of variance without applying correlation between follow eyes, as well as comparison of baseline and Week 12 and Week 24, and Weeks 12 and 24 with applying correlation between fellow eyes.

RV=retinal vasculitis; RVSS=retinal vasculitis severity scoring.
P<0.001 for the 24-week change of study indices by repeated measures analysis of variance without applying correlation between follow eyes, as well as comparison of baseline and Week 12 and Week 24, and Weeks 12 and 24 with applying correlation between fellow eyes.
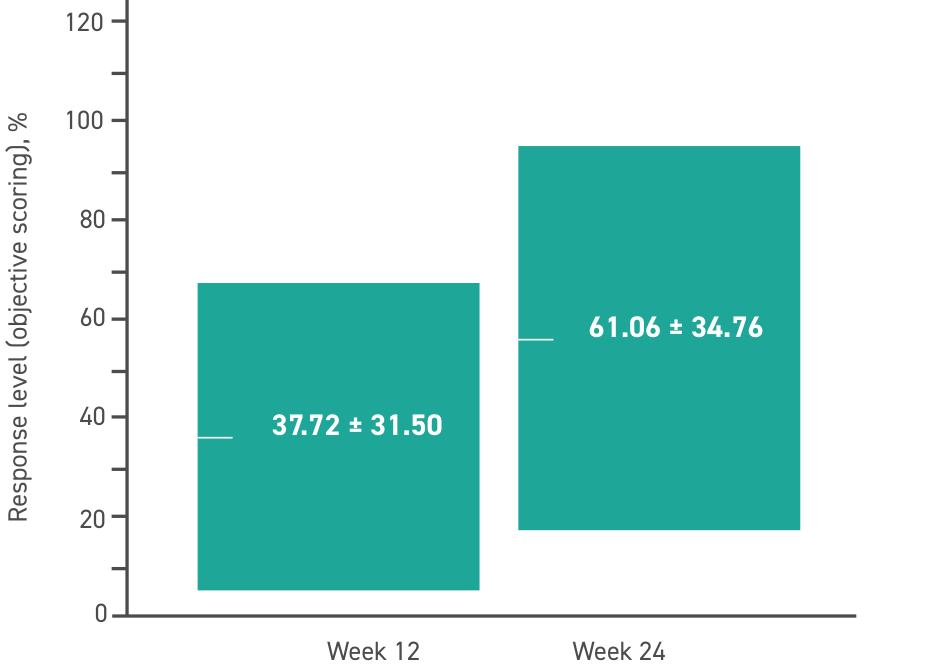
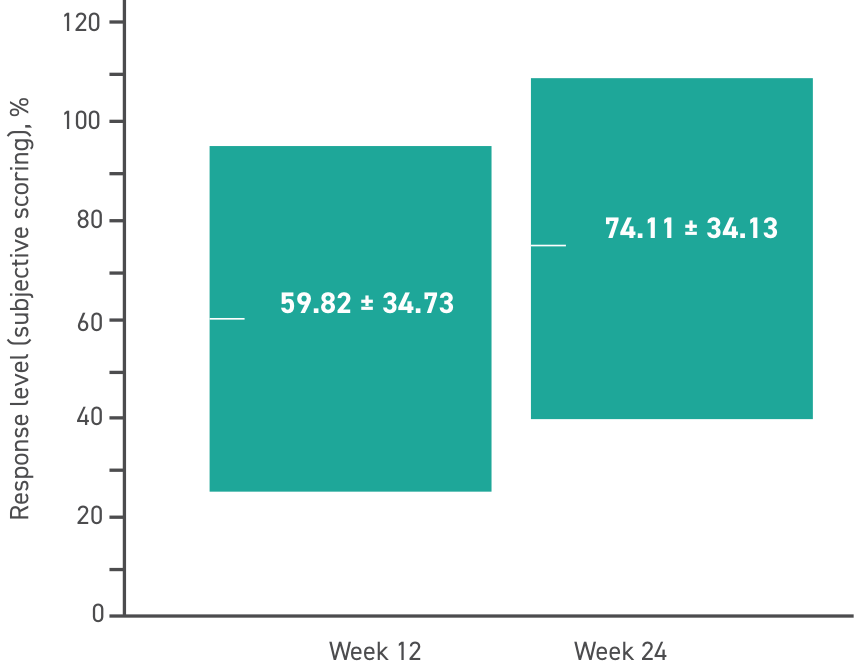
P<0.001 for the 24-week change of study indices by repeated measures analysis of variance without applying correlation between fellow eyes, as well as comparison of baseline and Week 12, baseline and Week 24, and Weeks 12 and 24 with applying correlation between fellow eyes.


Prolonged use of Acthar may produce cataracts, glaucoma, and secondary ocular infections. Monitor for signs and symptoms.
BCVA=best corrected visual acuity; IOP=intraocular pressure; logMAR=logarithm of the minimum angle of resolution.
*One patient (1 eye) was excluded because of shallow peripheral retinal detachments.
†Bolded values indicate statistical significance.
No concerning changes in laboratory values were found in any patient
No clinically significant increase in IOP was observed
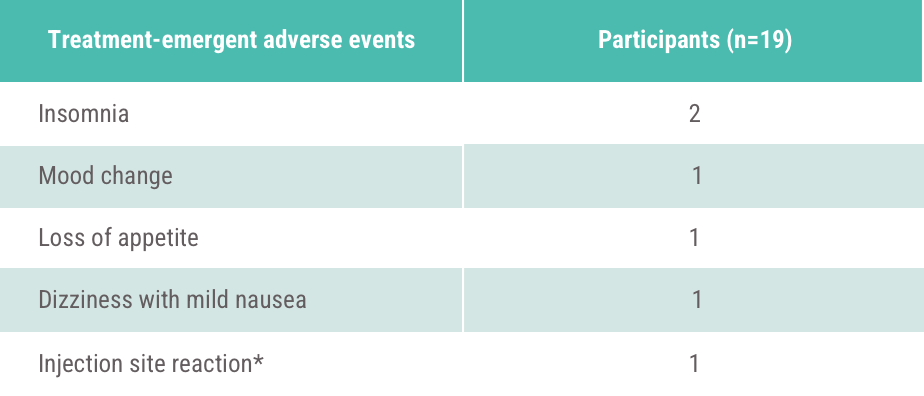
*One patient stopped Acthar Gel during study due to an adverse reaction (injection site reaction).

Start the referral process for your appropriate patients

See additional dosing information from clinical experience with Acthar Gel
Download and fill out the Doctor Notification Letter to share your patient's prescription with their other healthcare providers.
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References: