
Prescribe Acthar Gel now
Start the referral process for your appropriate patients
Fleischmann R, Furst DE, Connolly-Strong E, Liu J, Zhu J, Brasington R—Rheumatology and Therapy, 2020
Disclosure statement: Funding to support this study was provided by Mallinckrodt Pharmaceuticals.
To evaluate the efficacy, safety, and tolerability of Acthar Gel in patients with persistently active RA despite aggressive treatment with a glucocorticoid and 1 or 2 DMARD(s)
Part 1: 12-week, open-label treatment period
Part 2: 12-week, randomized, double-blind, placebo-controlled withdrawal period
DAS28-ESR=Disease Activity Score with 28 joint count and erythrocyte sedimentation rate; DMARD=disease-modifying antirheumatic drug; SC=subcutaneous.
Multicenter, two-part study

2x/week=2 times a week; LDA=low disease activity; R=randomization.
*The proportion of patients who achieved LDA (DAS28-ESR <3.2) at Week 12.
Primary endpoint
Selected secondary and exploratory endpoints
Safety endpoints evaluated by study period and throughout study
ACR20=American College of Rheumatology, 20% improvement; ACR50=American College of Rheumatology, 50% improvement; ACR70=American College of Rheumatology, 70% improvement; AEs=adverse events; CDAI=Clinical Disease Activity Index; FACIT-F=Functional Assessment of Chronic Illness Therapy-Fatigue; HAQ-DI=Health Assessment Questionnaire-Disability Index; WPAI=Work Productivity and Activity Impairment.
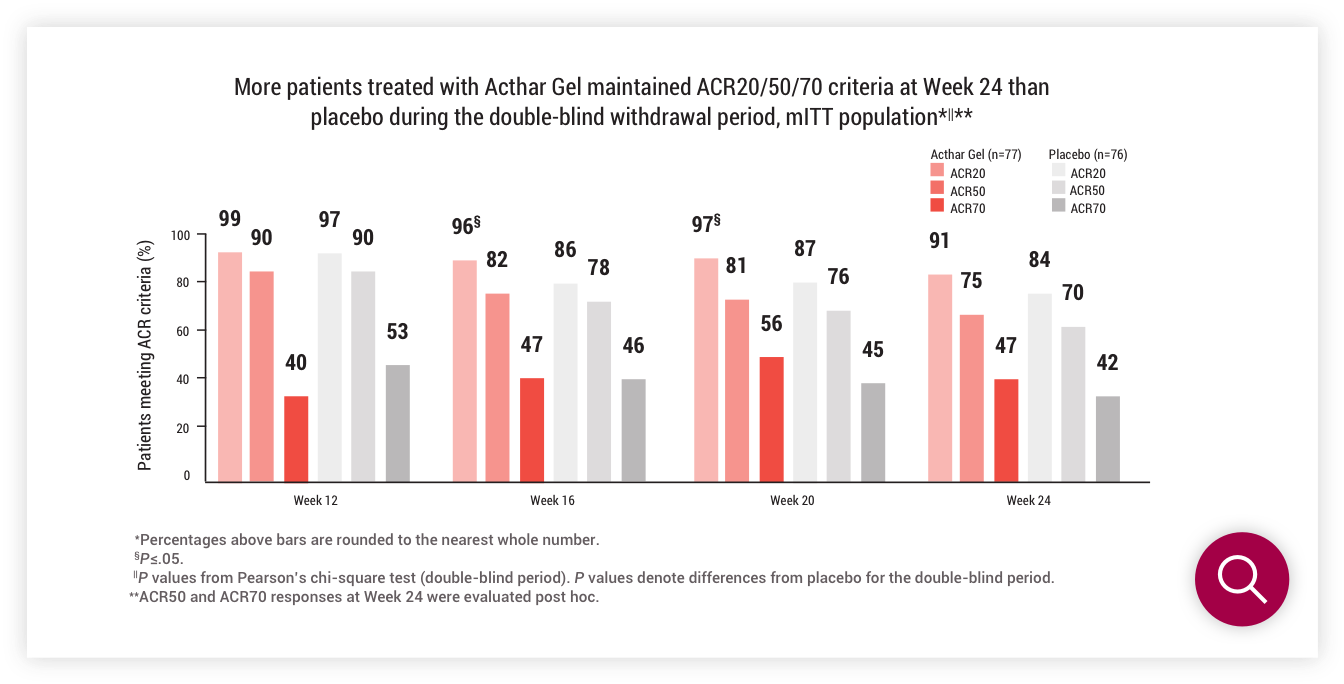
†ACR50 and ACR70 responses at Week 24 were evaluated post hoc.
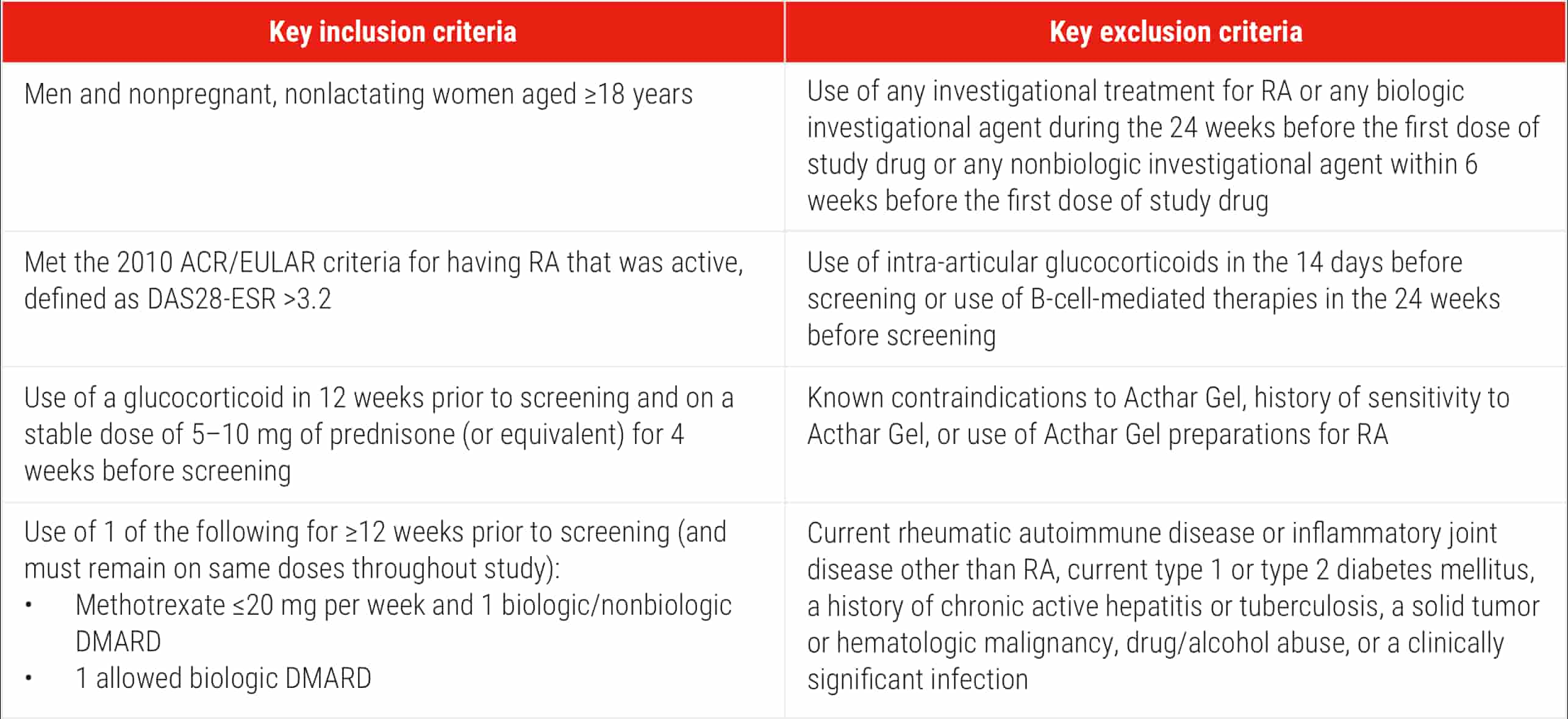

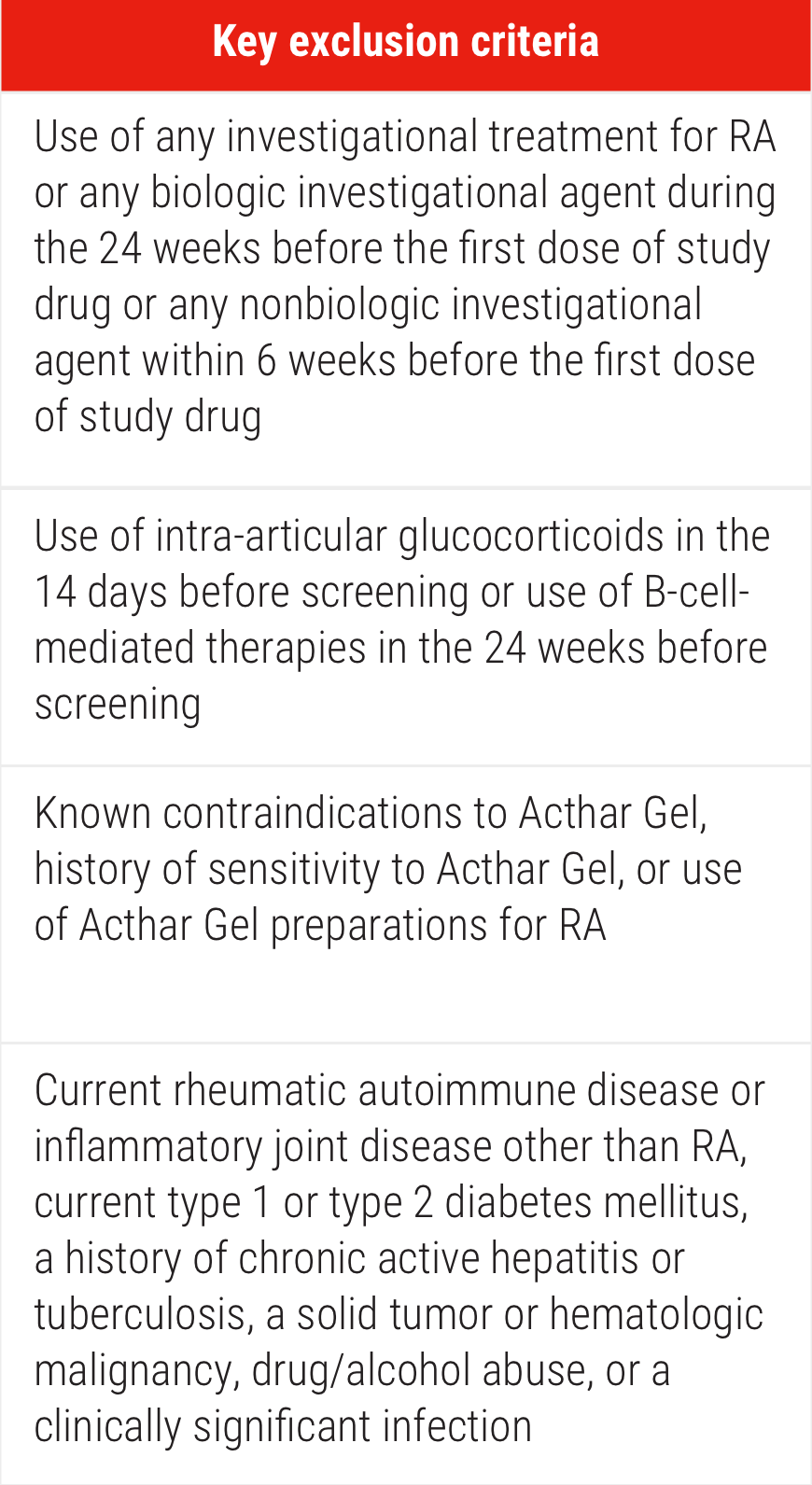
ACR=American College of Rheumatology; EULAR=European League Against Rheumatism.
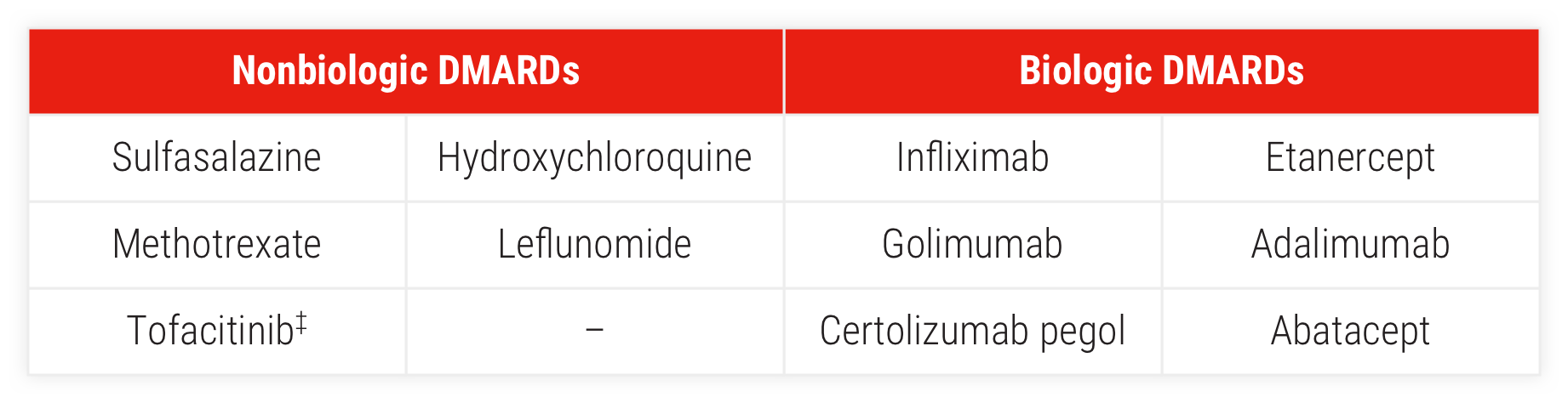
Most common (≥3% of patients) DMARDs included:
‡Targeted synthetic DMARD (tsDMARD).
§Prior DMARDs.
∥Concomitant DMARDs.
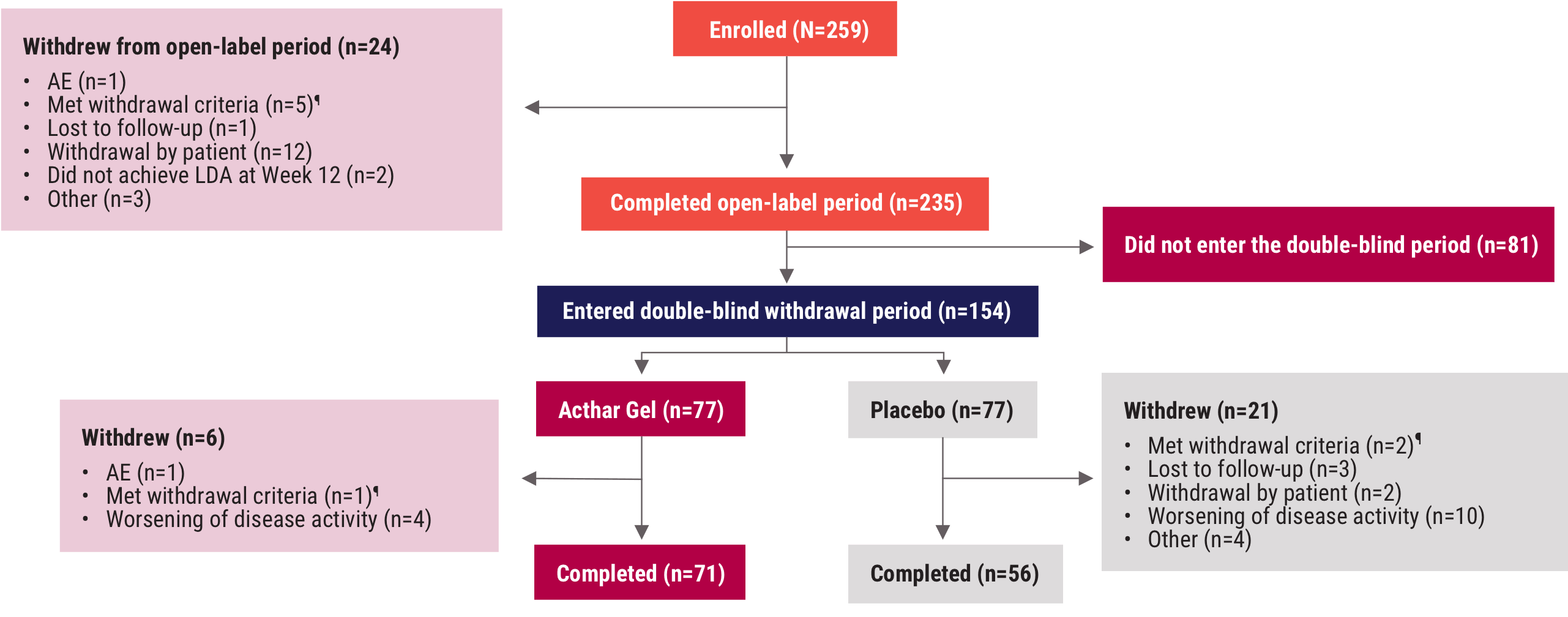
¶Patients met withdrawal criteria if they developed a condition that met any of the study exclusion criteria or failed to meet any inclusion criteria during the study that was not considered an AE, or if they were noncompliant.
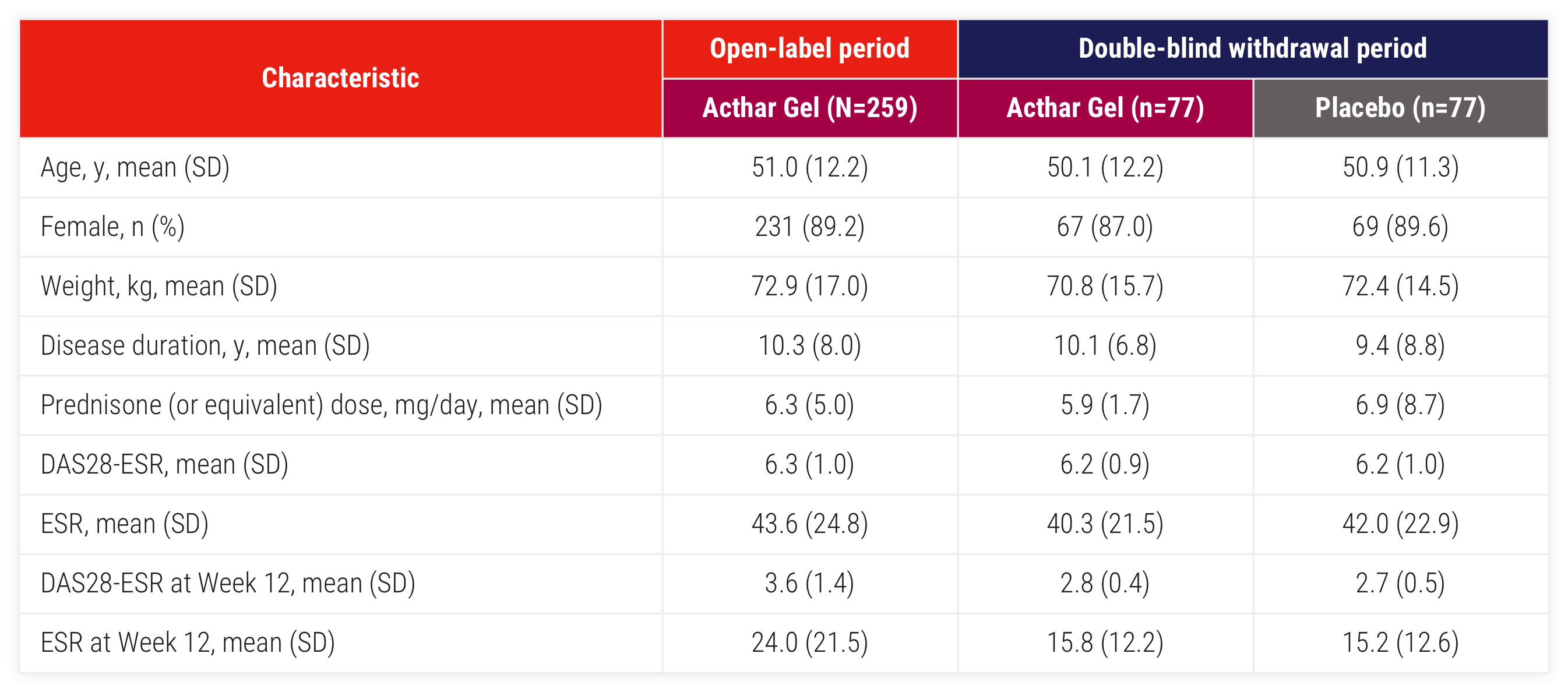
ESR=erythrocyte sedimentation rate; SD=standard deviation.
Patients in the study had persistently active disease despite treatment.
Efficacy results are presented for the modified intent-to-treat (mITT) population, which includes all patients who received ≥1 dose of study drug and contributed any efficacy data to the study.
Open-label treatment period (Part 1)
A majority of patients treated with Acthar Gel achieved LDA (DAS28-ESR <3.2) at Week 12
63% (n=163) of patients treated with Acthar Gel achieved LDA (DAS28-ESR <3.2) at Week 12 during the open-label period, mITT population*†
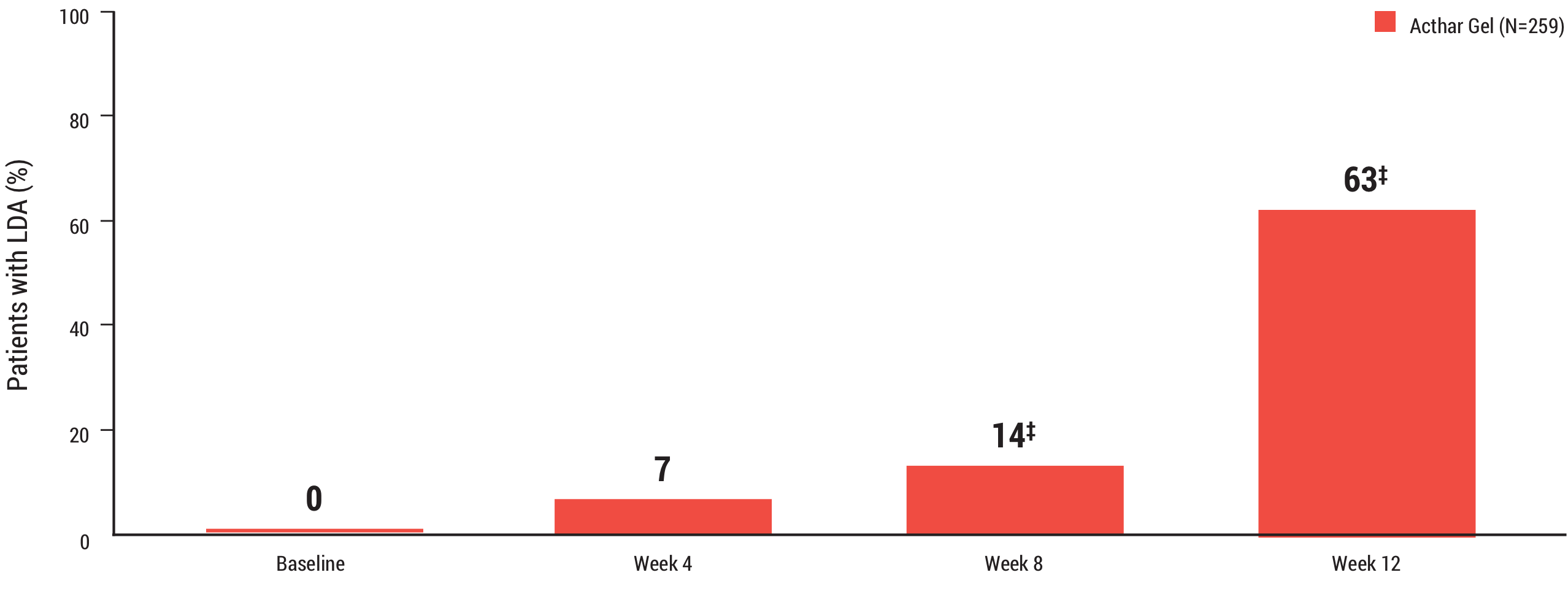
DAS28-ESR=Disease Activity Score with 28 joint count and erythrocyte sedimentation rate; LDA=low disease activity.
*Percentages above bars are rounded to the nearest whole number.
†P values from 1-sample binomial test (open-label period). P values denote differences from baseline for the open-label period.
‡P<.0001.
Open-label treatment period (Part 1)
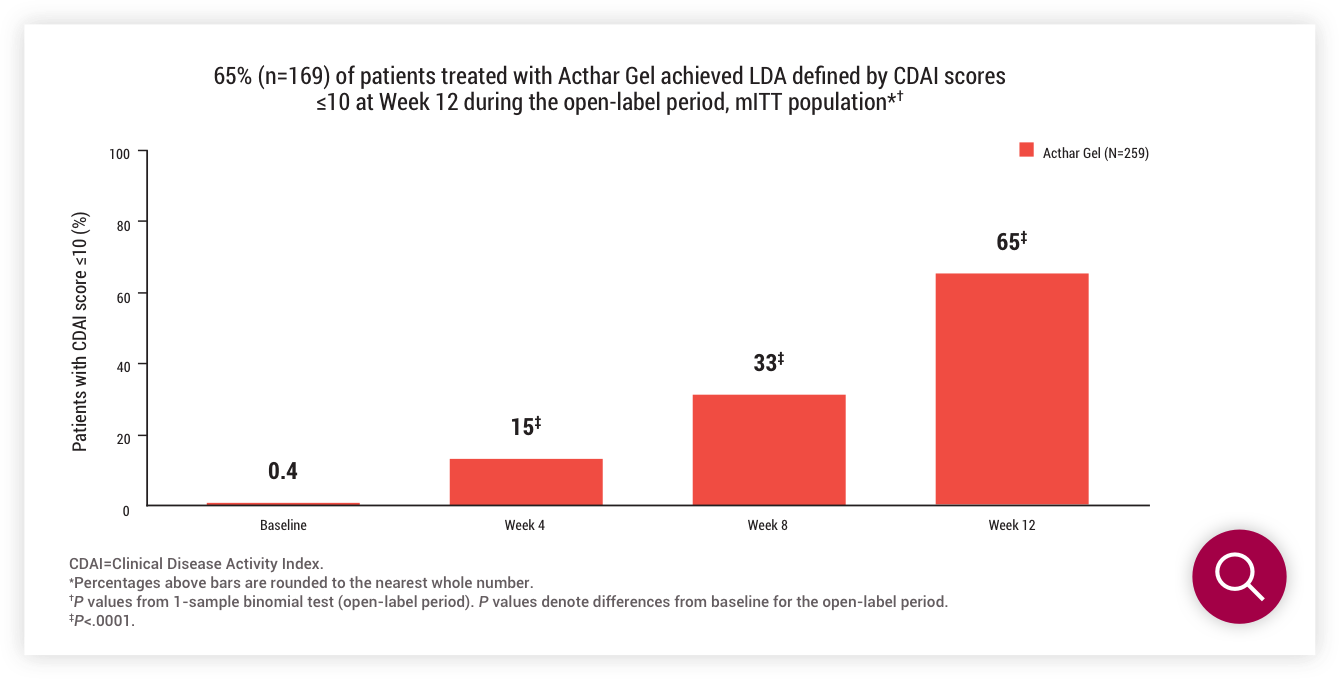
LDA defined by CDAI scores ≤10 at Week 12
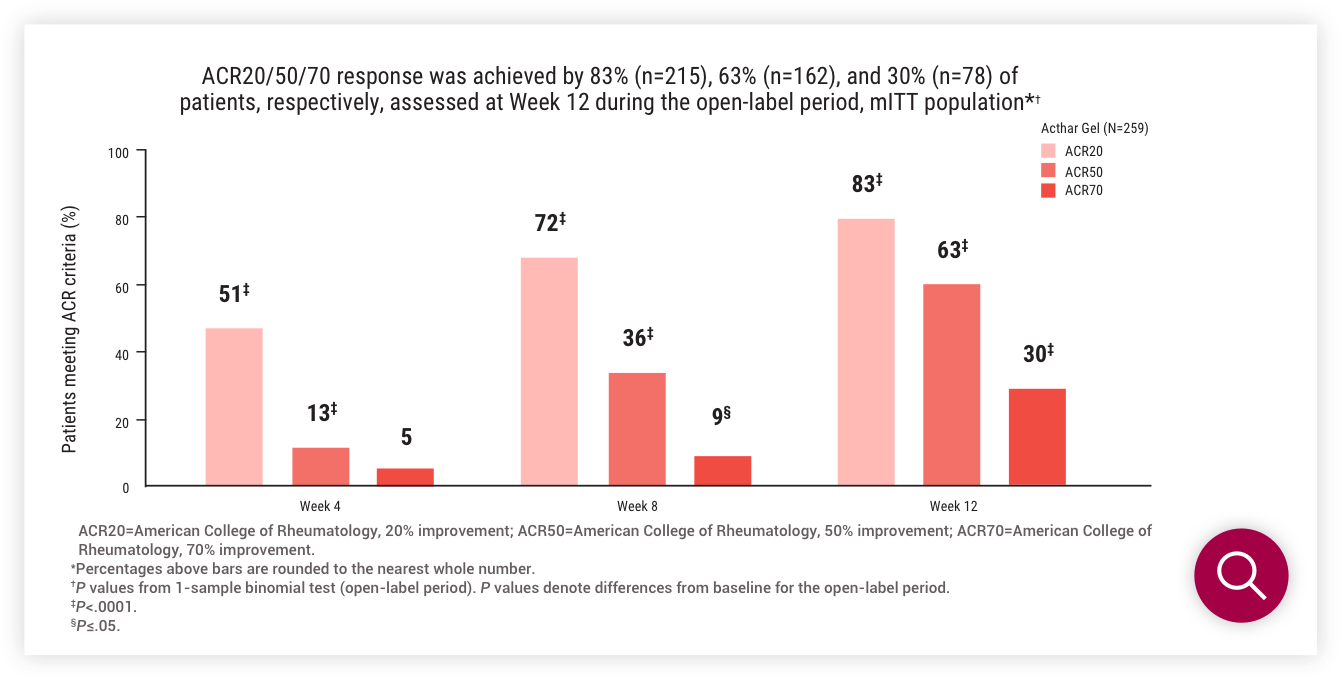
ACR20/50/70 at Week 12
Double-blind withdrawal period (Part 2)
LDA (DAS28-ESR <3.2) at Week 24
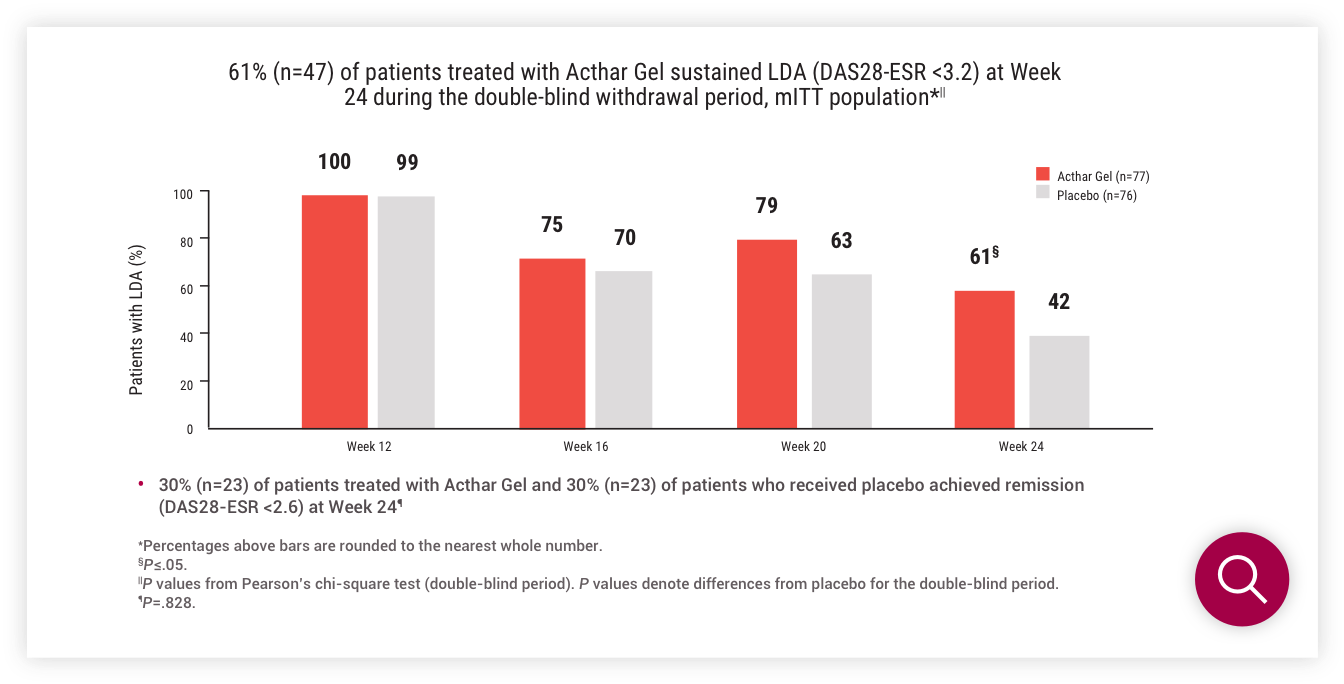
LDA (CDAI score ≤10) at Week 24
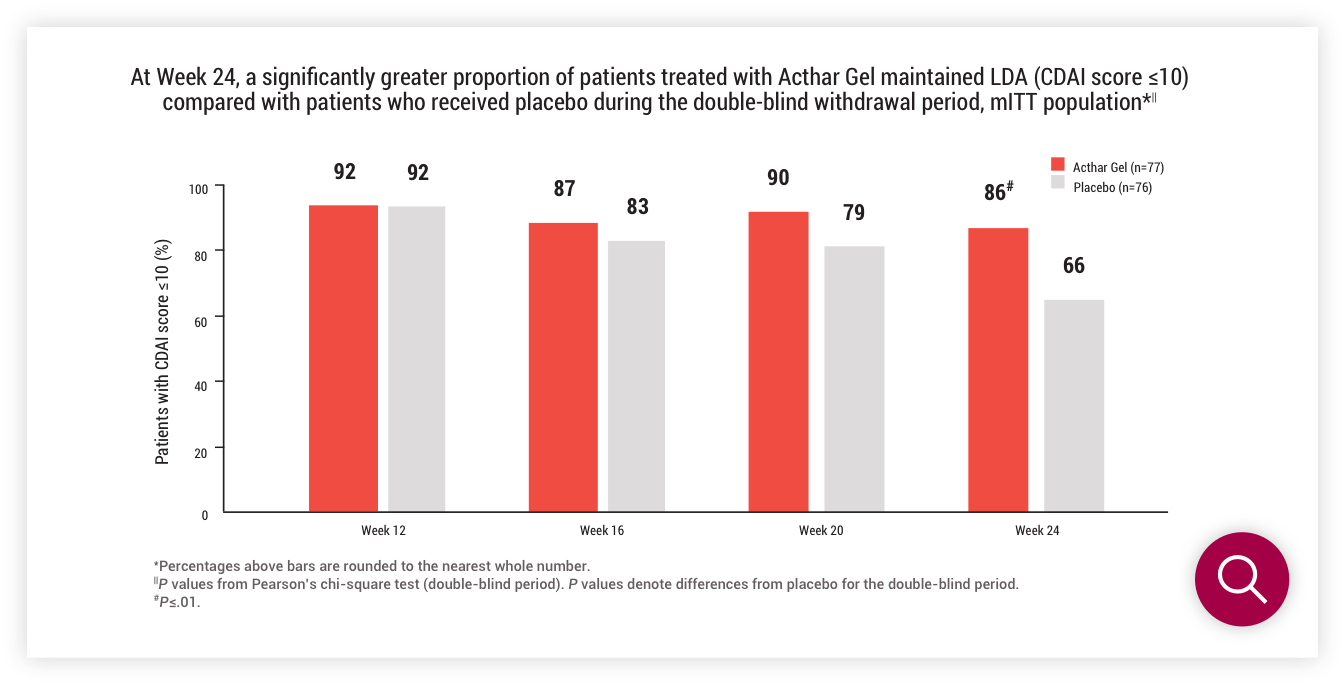
ACR20/50/70 criteria at Week 24
Open treatment period (Part 1)
Acthar Gel therapy was associated with significant improvements in swollen and tender joint counts and measures of fatigue (FACIT-F) and physical function (HAQ-DI) during the open-label period, mITT population (N=259)†
FACIT-F=Functional Assessment of Chronic Illness Therapy-Fatigue; HAQ-DI=Health Assessment Questionnaire-Disability Index; MCID=minimal clinically important difference; MID=minimal important difference; ND=not determined.
†P values from 1-sample binomial test (open-label period). P values denote differences from baseline for the open-label period.
‡‡P ≤.001.
§§P <.001.
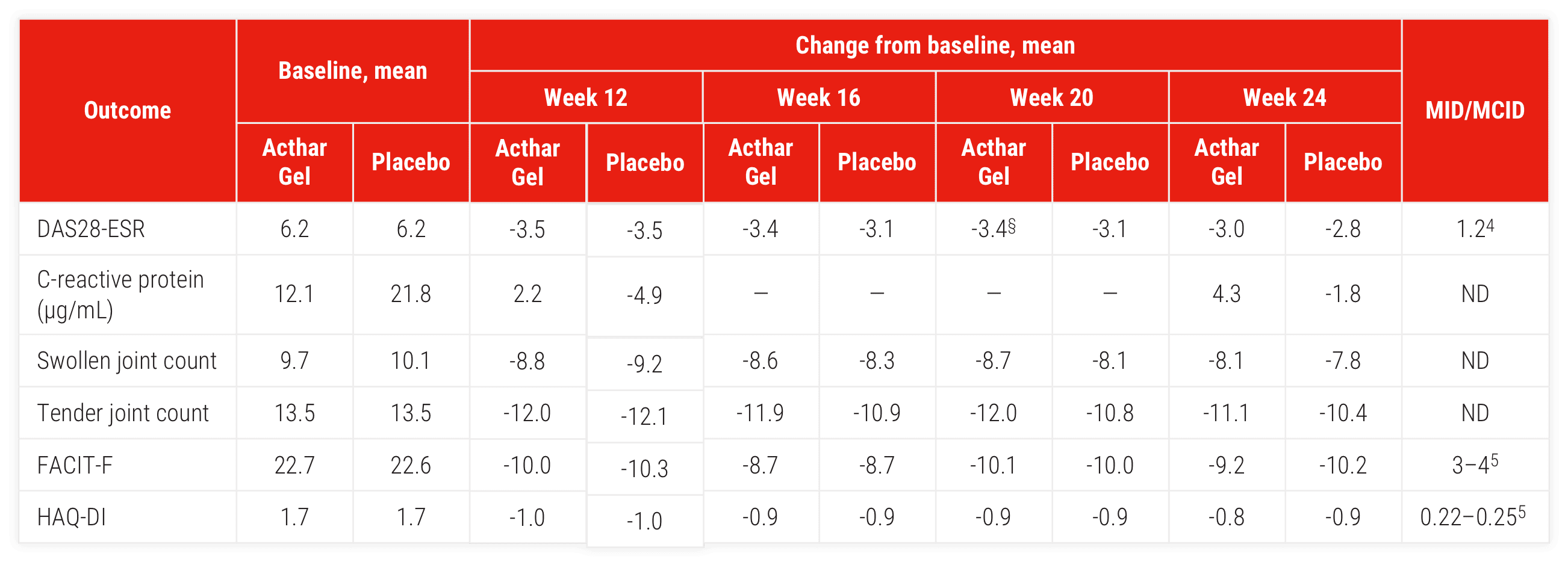
Double-blind withdrawal period (Part 2)
Acthar Gel therapy was associated with improvements in swollen and tender joint counts and measures of fatigue (FACIT-F) and physical function (HAQ-DI) during the double-blind withdrawal period, mITT population (Acthar Gel [n=77], placebo [n=76])∥
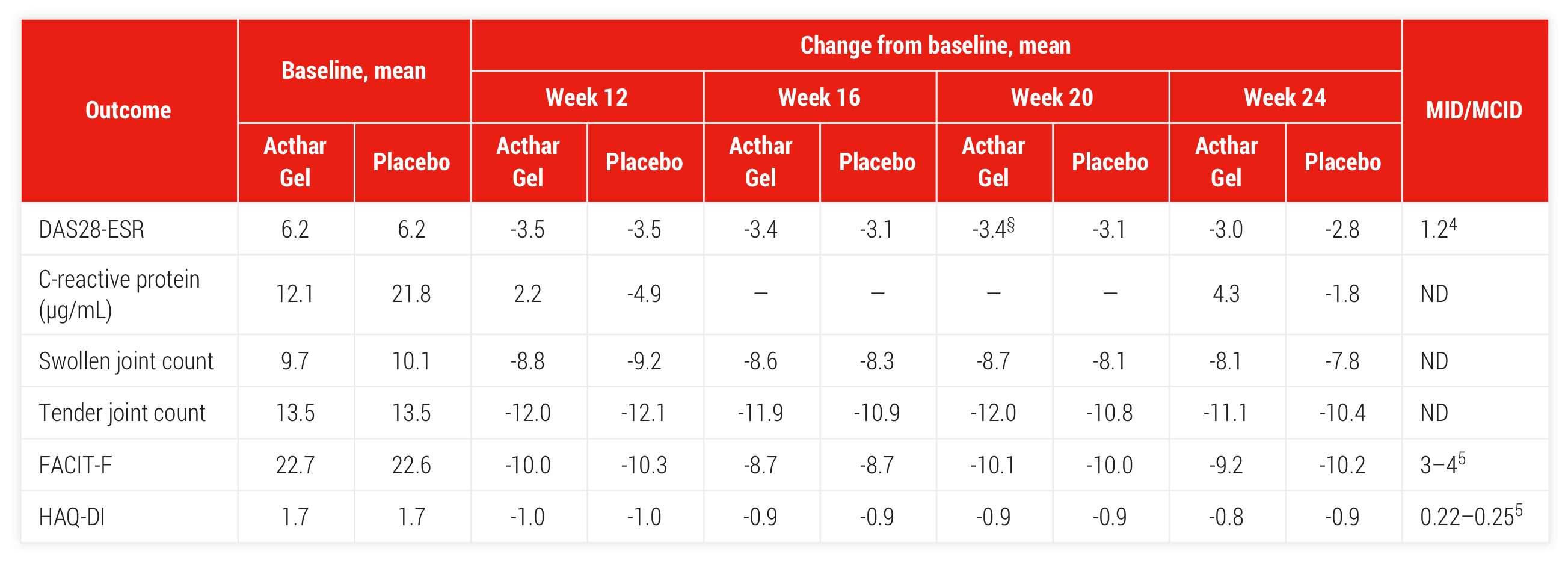
§P ≤.05.
∥P values from Pearson's chi-square test (double-blind period). P values denote differences from placebo for the double-blind period.
Summary of AEs, Safety Population

AE=adverse event.
*AEs reported in ≥1.5% of patients in part 1 or in either group in part 2.
†Refers to glycosylated hemoglobin values >6.5%.

Start the referral process for your appropriate patients

See additional dosing information from clinical experience with Acthar Gel
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References: