
Prescribe Acthar Gel now
Start the referral process for your appropriate patients
Hladunewich MA, Cattran D, Beck LH, Odutayo A, Sethi S, Ayalon R, Leung N, Reich H, Fervenza FC—Nephrology Dialysis Transplantation, 2014
Disclosure statement: Funding to support this study was provided by Mallinckrodt Pharmaceuticals.
To assess the safety and efficacy of Acthar Gel in 20 adults (>18 years) with biopsy-proven iMN with proteinuria due to nephrotic syndrome
Primary outcomes included:
Secondary endpoints included the proportion of patients who achieved:
iMN=idiopathic membranous nephropathy.
*Proteinuria <0.3 g/day.
†Reduction in proteinuria by >50% with a final urine protein <3.5 g/day, but >0.3 g/day.
‡Reduction in proteinuria by <50%, or worsening of proteinuria.
At baseline, the mean age of the study population was 51 years and the median disease latency was 14 months. Thirteen of 20 patients had not received previous immunosuppressive agents.
§Significant renal insufficiency was defined as estimated glomerular filtration rate (eGFR) ≥40 mL/min/1.73 m2 while taking RAS blockade.
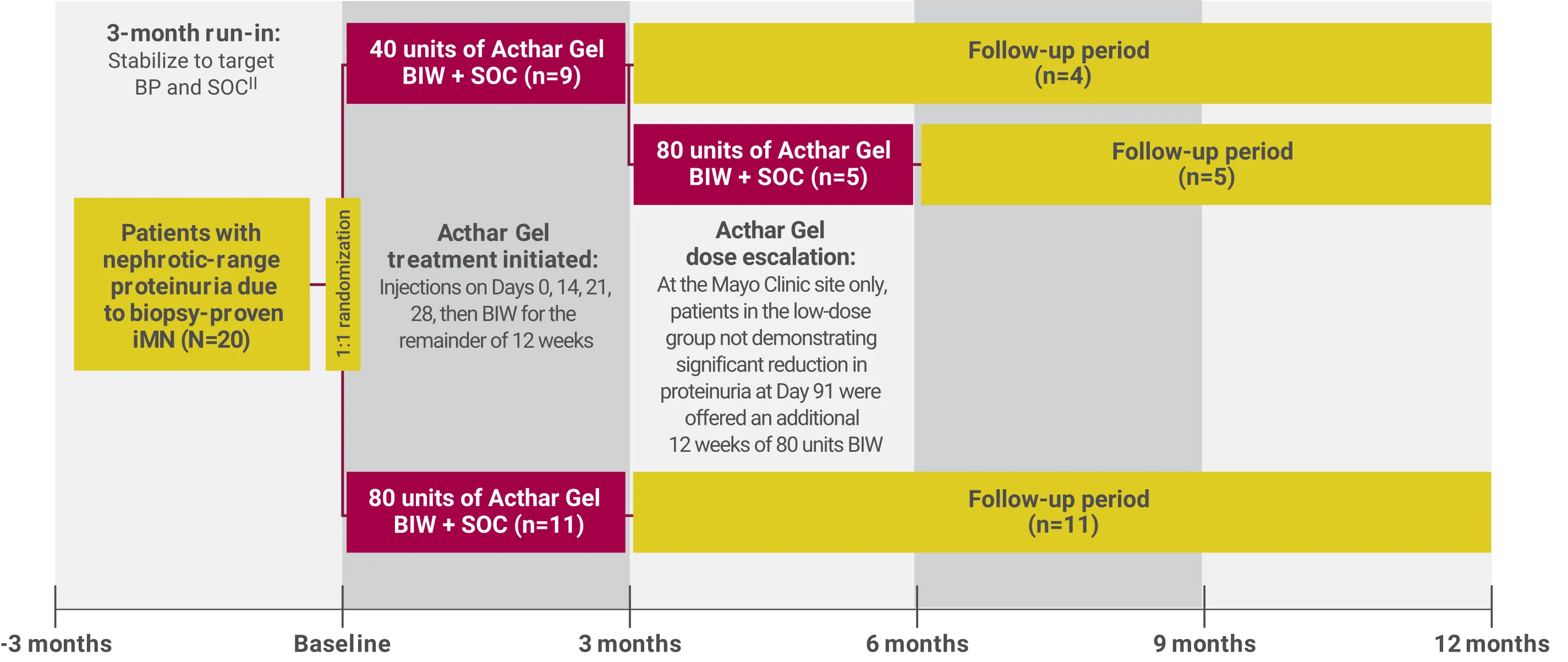
BIW=twice weekly; SOC=standard of care.
||SOC included statin therapy at the maximum recommended dose, dietary counseling to maintain a low-salt diet (2–3 g/day), and a target dietary protein intake of 0.8 g/kg ideal body weight/day of high-quality protein.
Significant improvement in mean proteinuria was observed at completion of Acthar Gel treatment and 12-month follow-up1
Mean proteinuria improved and was reduced significantly over 12 months, even after Acthar Gel treatment was completed

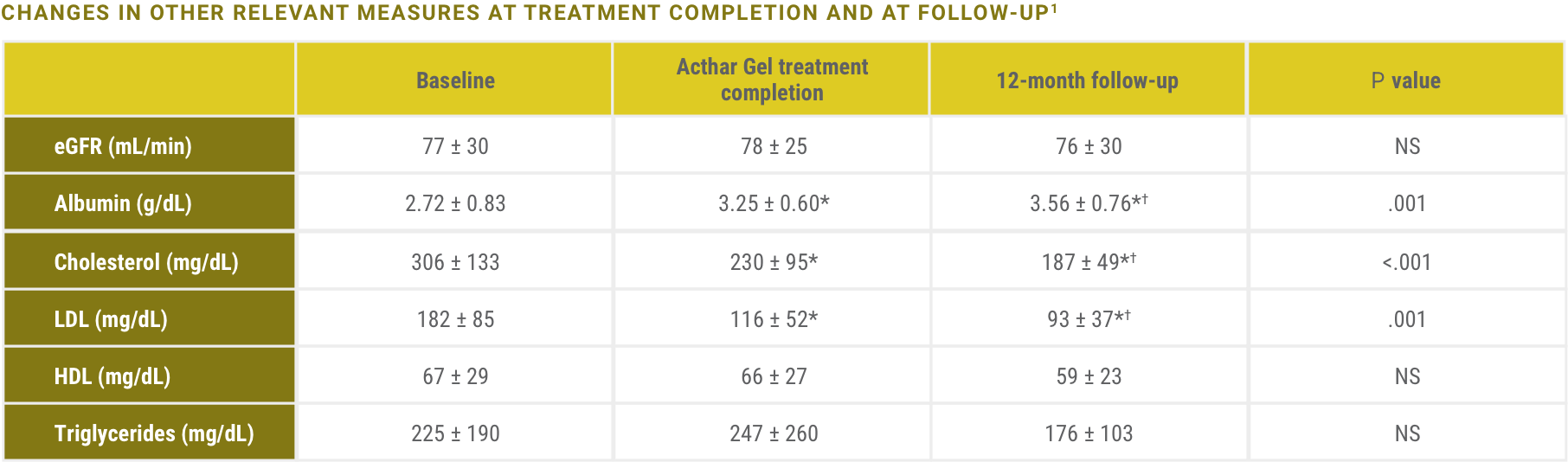
Acthar Gel is indicated to reduce proteinuria in nephrotic syndrome.2 The impact of Acthar Gel on other relevant lab values has not been more formally assessed and is included here for clinical context only.
eGFR=estimated glomerular filtration rate; HDL=high-density lipoprotein; LDL=low-density lipoprotein; NS=not significant.
*P<.05 versus baseline.
†P<.05 versus Acthar Gel treatment completion.
Patients experienced a greater than 50% reduction in proteinuria1
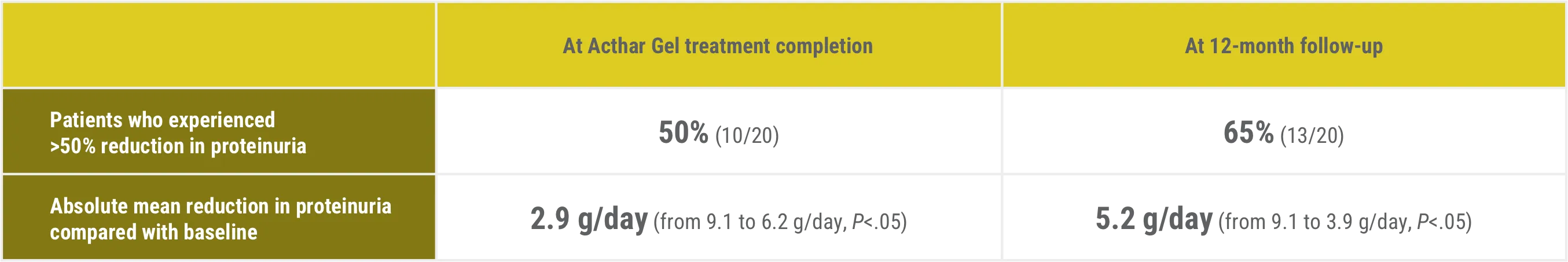
‡Complete remission was defined as proteinuria <0.3 g/day. Partial remission was defined as a reduction in proteinuria by >50% with a final urine protein <3.5 g/day, but >0.3 g/day. No response was defined as a reduction in proteinuria by <50%, or worsening of proteinuria.
§One patient experienced significant improvement in proteinuria, but did not meet criteria for partial remission.
||Of these patients, 2 were given alternate immunosuppression prior to completion of the trial and were excluded from 12-month follow-up data.
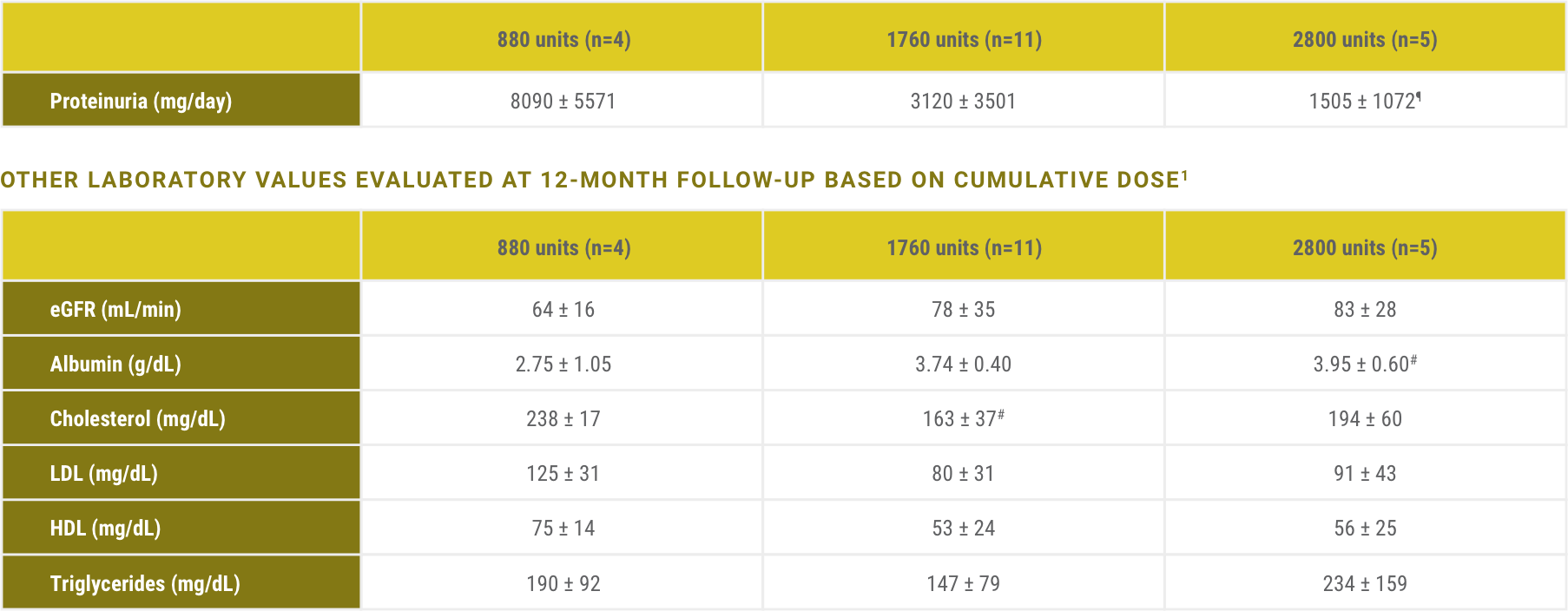
Acthar Gel is indicated to reduce proteinuria in nephrotic syndrome. The impact of Acthar Gel on other relevant lab values has not been more formally assessed and is included here for clinical context only.
¶P<.05 for trend across groups.
#P<.05 versus 880 units.
No patient discontinued Acthar Gel therapy due to treatment-related adverse events
The following adverse events occurred during the year of follow-up, but not all cases were clearly related to the therapy: tremulousness (n=3), hoarseness (n=2), dizziness (n=5), muscle aches or pain (n=5), headaches (n=5), gastrointestinal symptoms (n=7), blurred vision (n=2), and generalized weakness or fatigue (n=9).
Transient increases to a blood glucose level ≥130 mg/dL occurred in 5 patients. One patient sustained a clinically important glucose increase and required dietary treatment, which was improved with weight loss.

Start the referral process for your appropriate patients

See additional dosing information from clinical experience with Acthar Gel
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References: