
Prescribe Acthar Gel now
Start the referral process for your appropriate patients
Alhamad T, Manllo Dieck J, Younus U, et al—Transplantation, 2019
Disclosure statement: Funding to support this study was provided by Mallinckrodt Pharmaceuticals.
To investigate the use of Acthar Gel in kidney transplant recipients with de novo or recurrent FSGS resistant to TPE and rituximab
TPE=therapeutic plasma exchange.
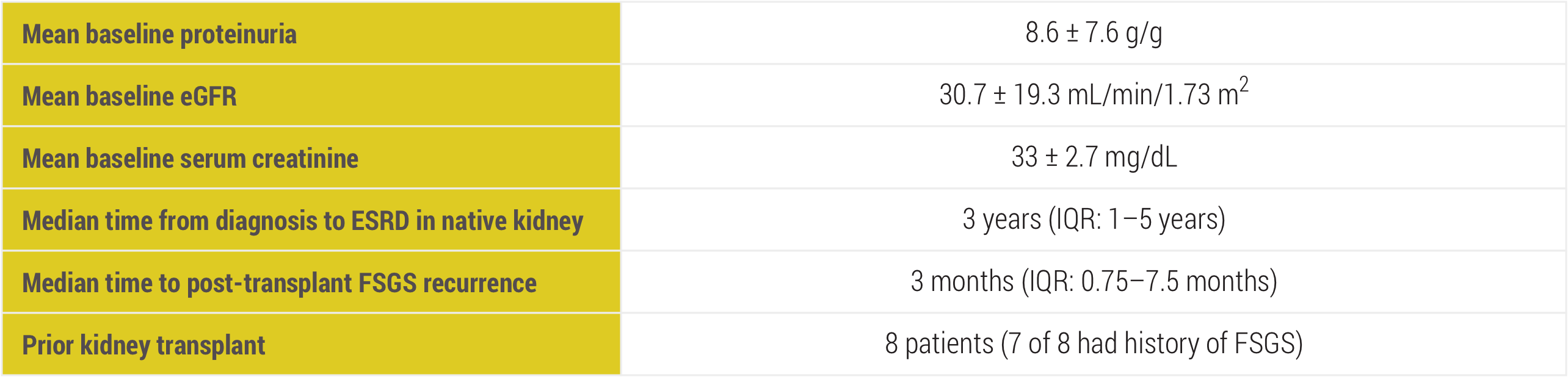
eGFR=estimated glomerular filtration rate; ESRD=end-stage renal disease; IQR=interquartile range.

ACEi=angiotensin-converting enzyme inhibitor; ARB=angiotensin receptor blocker; CNI=calcineurin inhibitor; MMF=mycophenolate mofetil.
*Two patients did not receive any ACEi or ARB.
†Dosage and frequency should be individualized according to the medical condition, severity of disease, and initial response of the patient.
TPE=therapeutic plasma exchange
Significant reduction in proteinuria (N=20)1
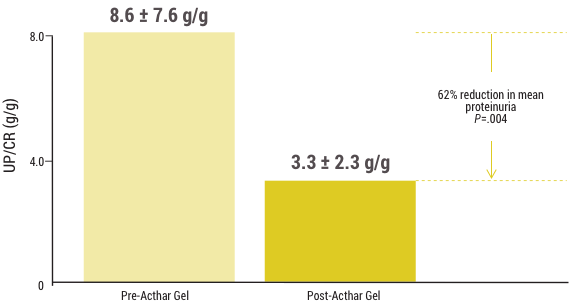
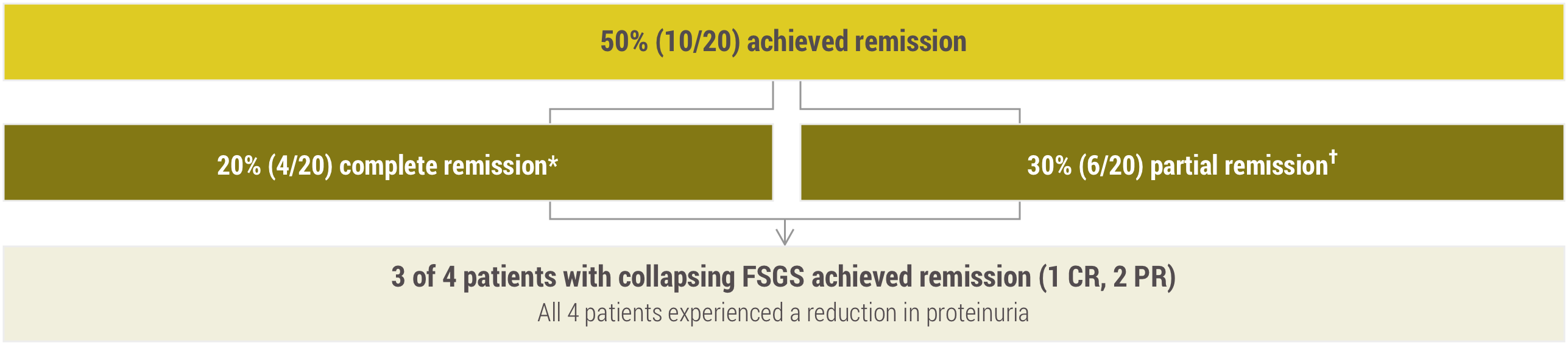
Some post-transplant FSGS patients who were treated achieved remission1
Stable renal function was observed following Acthar Gel therapy1*†
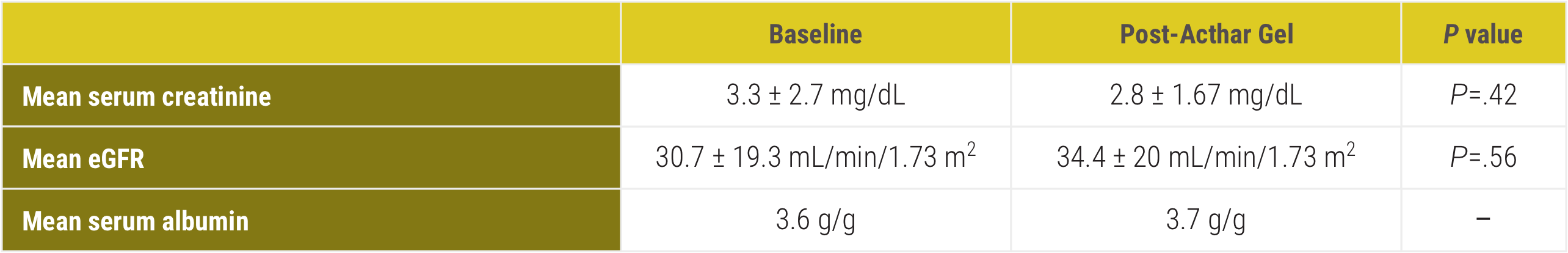
CR=complete remission; eGFR=estimated glomerular filtration rate; PR=partial remission; UP/CR=urine protein/creatinine ratio.
*Complete remission: decrease in proteinuria <1 g/g with stable kidney function (creatinine within 30% from baseline).
†Partial remission: decrease in proteinuria between 1 and <3.5 g/g with stable kidney function (creatinine within 30% from baseline).
Other observations reported for patients with post-transplant FSGS1
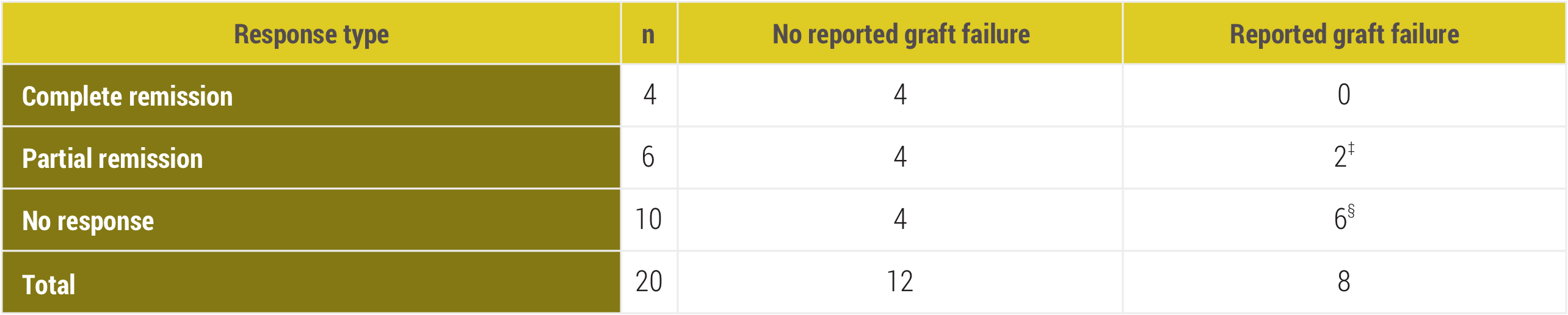
Graft status (2012-2016 observation period)
Therapeutic plasma exchange (TPE)

‡Graft failures reported 27 and 36 months after completion of Acthar Gel treatment.
§Five graft failures were attributed to recurrent or de novo FSGS despite the use of Acthar Gel; 1 was complicated by cytomegalovirus disease, and 1 by JC nephropathy.

Start the referral process for your appropriate patients

See additional dosing information from clinical experience with Acthar Gel
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References: