
Prescribe Acthar Gel now
Start the referral process for your appropriate patients
Fernandez AP, Gallop J, Polly S, Khanna U—Rheumatology (Oxford), 2023
Disclosure statement: Funding to support this study was provided by Mallinckrodt Pharmaceuticals. An author of this study has a financial and/or employment relationship with Mallinckrodt Pharmaceuticals.
To assess the efficacy and safety of Acthar Gel over 6 months in 15 patients with refractory cutaneous DM

Patients attended follow-up visits for full physical examinations and clinical scoring assessments at baseline, 1 month, 3 months, and 6 months after Acthar Gel initiation.
*The CDASI is a valid and responsive measure used to characterize the severity of cutaneous dermatomyositis and detect improvement in disease activity. PGA is an overall rating of disease severity that is completed on a 10-cm continuous visual analog scale, ranging from 0 (no evidence of disease) to 10 (extremely severe disease).2

CADM=clinically amyopathic dermatomyositis; CDASI-A=Cutaneous Dermatomyositis Disease Area and Severity Index Activity.
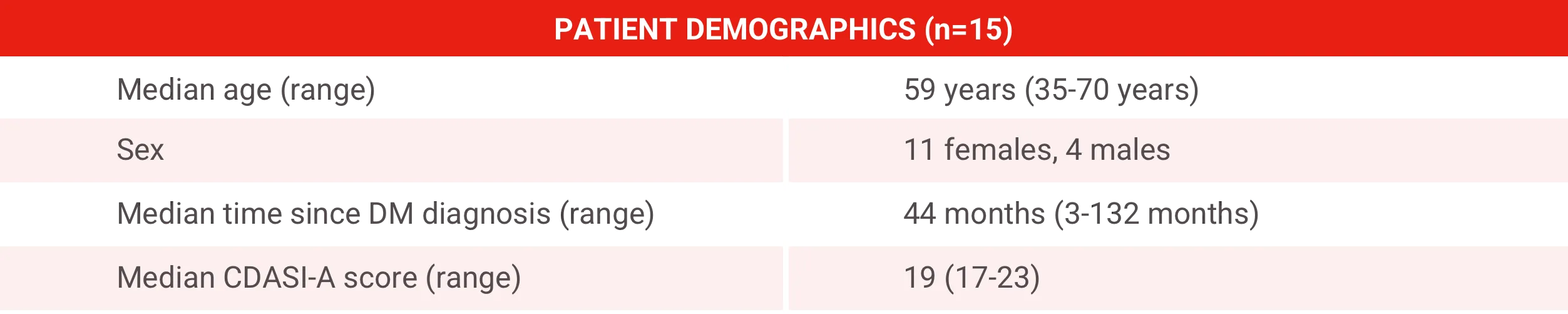
All patients previously received systemic medications and were receiving a median of 2 systemic medications at the time of enrollment.
Limitations of this study include a relatively small sample size, the lack of a placebo arm, and the open-label design, which may introduce physician and patient bias.
Because 3 patients withdrew shortly after enrollment and were not included in analyses, some degree of selection bias cannot be entirely ruled out. Importantly, however, objective correlation between clinical photographs and assessment scores was noted for all patients.
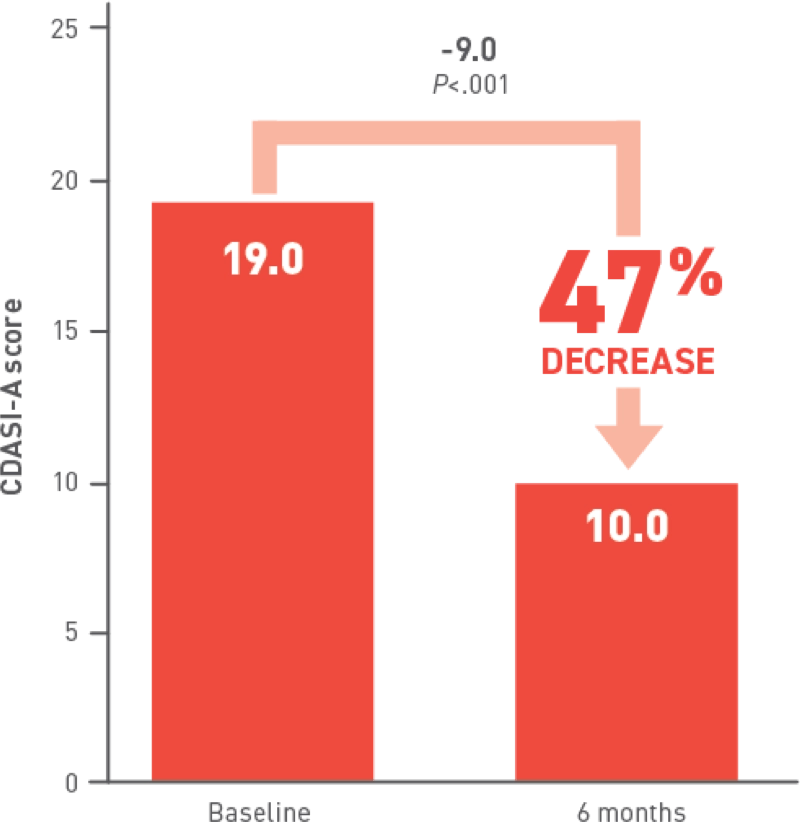

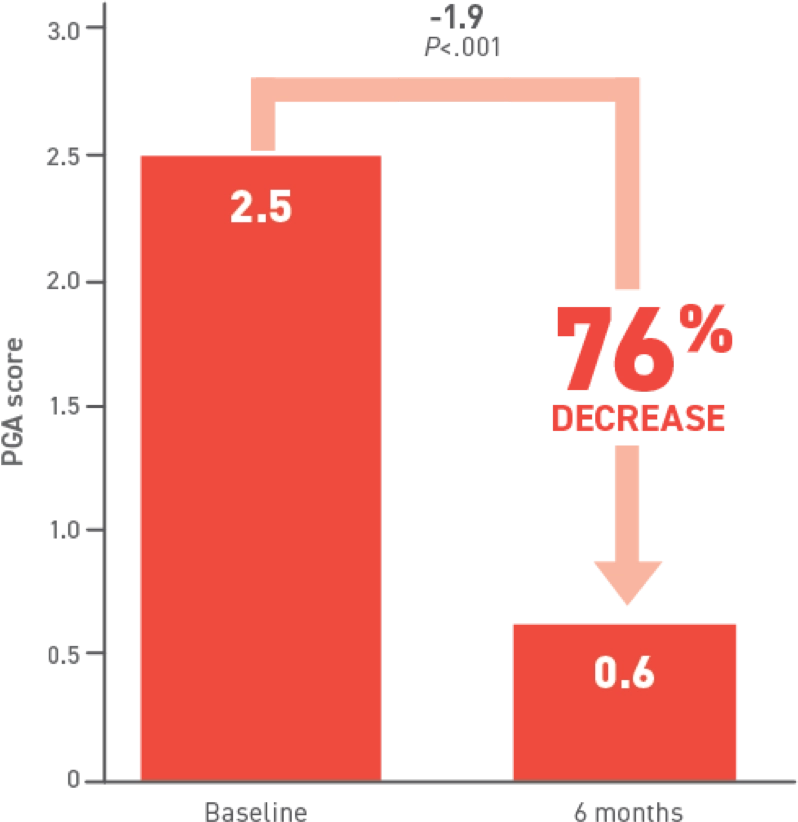
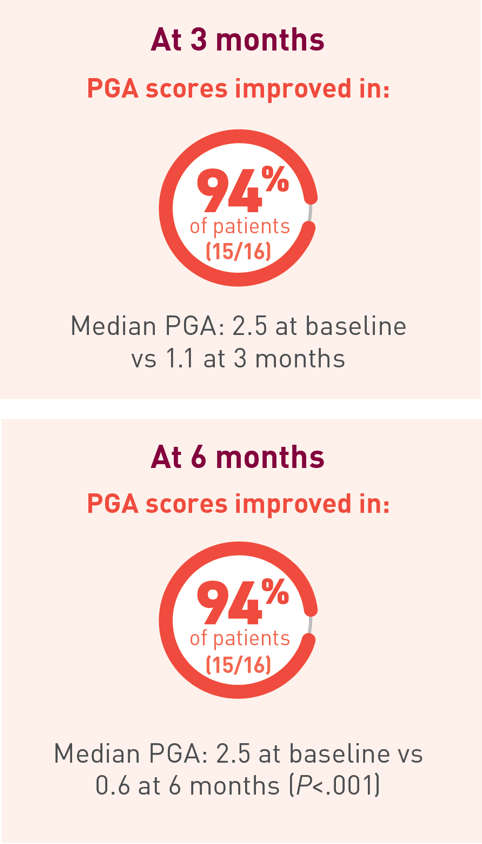
At 6 months, patients receiving Acthar Gel saw
improvements in all typical DM cutaneous
lesions,
including
alopecia and cutaneous erythematous inflammation
involving:

After 6 months of treatment with Acthar Gel, physician-assessed PhGSS, MITAX (a component of the MDAAT), and CAT scores showed statistically significant improvements compared with baseline



Analysis of MMT-8 scores in classic DM patients (n=7) revealed no significant changes at 3 or 6 months compared with baseline.




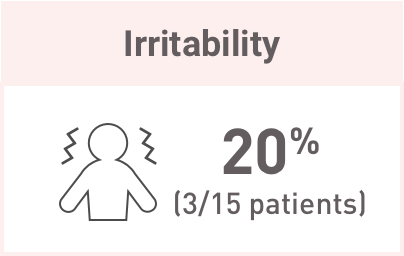
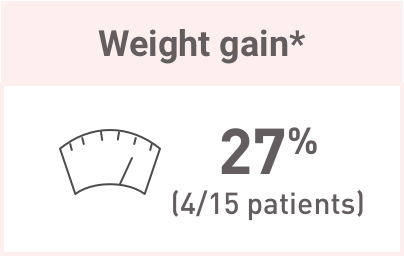
These are common glucocorticoid-associated AEs and may be related to Acthar Gel-induced endogenous cortisol production.
At baseline and 6 months, there were no significant abnormalities seen in evaluated laboratory parameters. Although median baseline values were within normal range at 6 months, there were:

Start the referral process for your appropriate patients
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References:
References: